[ad_1]
Isolation and characterization of MPs derived from apoptotic HPCs
Throughout apoptosis, cells can launch small MPs referred to as apoMPs (< 1 μm in diameter) [21]. With a view to receive a helpful quantity of apoMPs from apoptotic HPCs, we used completely different deadly doses of doxorubicin (Extra file 2: Fig. S1A) to deal with HPCs (WB-F344 cell line) and the launched MPs had been then remoted by centrifugation steps (Extra file 2: Fig. S1B) [22]. It has beforehand been proven that when tumor cells had been handled with doxorubicin, the doxorubicin was packaged into the launched MPs, and due to the fluorescent nature of doxorubicin, MPs encapsulating doxorubicin may very well be clearly noticed by fluorescence microscopy [22]. The launched apoMPs had been additionally noticed by fluorescence microscopy in our research. We discovered that WB-F344 cells handled with 100 µg/ml of doxorubicin launched a substantial quantity of apoMPs (apoHPC-MPs) (Extra file 2: Fig. S1C). Move cytometry additional confirmed that 2 × 107 WB-F344 cells launched about 1 × 106 apoHPC-MPs (Extra file 2: Fig. S2A, S2B). These apoHPC-MPs had a membrane construction and had been ~800 nm in measurement, as detected by transmission electron microscopy (TEM; Extra file 2: Fig. S2C). The sizes had been confirmed by dynamic gentle scattering (DLS) evaluation (Extra file 2: Fig. S2D). DLS evaluation additionally revealed that apoHPC-MPs displayed zeta potentials of ~ − 17 mV (Extra file 2: Fig. S2E). Thus, throughout apoptosis, HPCs can launch giant numbers of apoHPC-MPs.
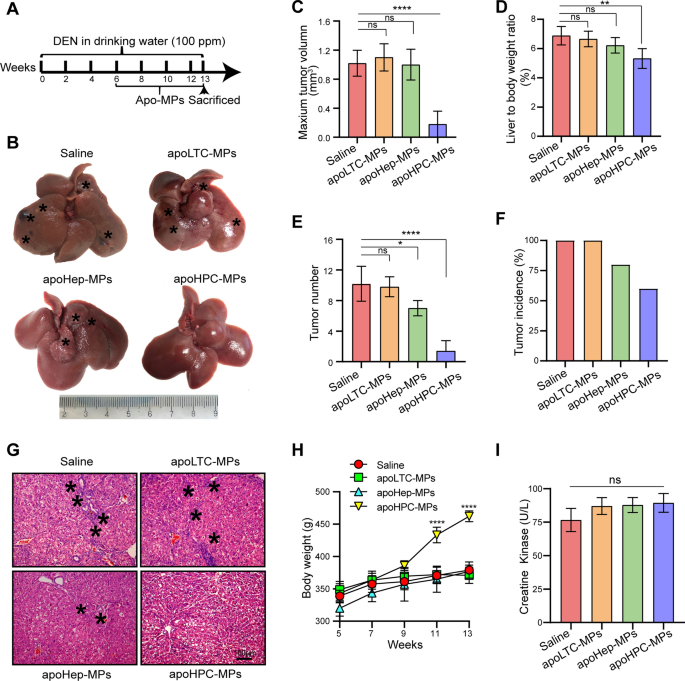
MPs derived from apoptotic HPCs stop hepatocarcinogenesis in a major rat HCC mannequin
To check whether or not apoMPs can inhibit hepatocarcinogenesis, it’s important to determine which mobile varieties are appropriate sources of apoMPs. In addition to the apoHPC-MPs described above, which had been produced from WB-F344 cells, we additionally produced apoMPs from the hepatocyte cell line BRL (apoHep-MPs) and the liver tumor cell line RH35 (apoLTC-MPs) by remedy with 100 µg/ml of doxorubicin. The apo-MPs had been noticed by fluorescence microscopy (Extra file 2: Fig. S2F). ApoHPC-MPs, apoHep-MPs and apoLTC-MPs had an analogous irregularly spherical morphology as noticed by TEM (Extra file 2: Fig. S2C). Additionally they had related diameters of ~800 nm (Extra file 2: Fig. S2D) and related zeta potentials of ~ − 17 mV (Extra file 2: Fig. S2E). To check whether or not these apoMPs have related anticancer results, we employed the diethylnitrosamine (DEN)-induced major HCC mannequin in Sprague Dawley rats. 6 weeks of oral DEN remedy confirmed the apparent HPC activation (Extra file 2: Fig. S3), HPCs had been labeled with an antibody in opposition to CK7 [23], and in our earlier research, we discovered that the activation and malignant transformation of HPCs promote hepatocarcinogenesis and HCC recurrence [9,10,11,12]. Thus, the rats had been intrasplenically administered with 40 µg of apoLTC-MPs, apoHep-MPs, apoHPC-MPs or saline after 6 weeks of oral DEN remedy. Rats had been injected with MPs or saline twice per week for 7 weeks, and oral DEN remedy was continued on the identical time. On week 13 of DEN remedy, the rats had been sacrificed (Fig. 1A). We then assessed the therapeutic efficacy of apoMPs within the rat HCC major mannequin. In contrast with saline management, apoLTC-MPs or apoHep-MPs, the apoHPC-MPs successfully inhibited tumorigenesis in DEN-exposed rats (Fig. 1B). This was additionally evidenced by the utmost tumor quantity (Fig. 1C), liver-to-body weight ratio (Fig. 1D), tumor quantity (Fig. 1E) and tumor incidence (Fig. 1F). H&E staining confirmed that livers from rats handled with apoHPC-MPs had a decreased inflammatory response and a transparent tissue construction (Fig. 1G). In addition to, we discovered that apoHPC-MPs ameliorated the load lack of rats throughout hepatocarcinogenesis (Fig. 1H). Due to the identified toxicity of doxorubicin in coronary heart [22], we additionally examined whether or not the apoMPs have unwanted effects on coronary heart. Notably, we didn’t observe poisonous results of apoHPC-MPs in coronary heart, as proven by serological evaluation (creatine kinase) and H&E staining (Fig. 1I and Extra file 2: Fig. S4). ApoHPC-MPs additionally had no unwanted effects in different main organs, as evidenced by H&E staining of different tissues (Extra file 2: Fig. S4). Taken collectively, these outcomes present that apoHPC-MPs have a lot better anticancer efficacy than apoLTC-MPs and apoHep-MPs with out typical unwanted effects in a rat major HCC mannequin.
MPS derived from apoptotic HPCs stop hepatocarcinogenesis in a major rat HCC mannequin. A Diagram of the remedy schedule within the rat HCC mannequin. After 6 weeks of oral remedy with diethylnitrosamine (DEN), Sprague Dawley rats had been intrasplenically injected with 40 µg of apoHPC-MPs, apoLTC-MPs or apoHep-MPs in 200 µl saline or 200 µl of clean saline. Injections had been administered twice each week for 7 weeks. Oral DEN remedy was additionally continued throughout this time. After 13 weeks, the rats had been sacrificed to watch the event of hepatocellular carcinoma (HCC). B Consultant pictures of rat livers from the indicated teams. Typical tumor nodes are marked by the asterisks. C The utmost tumor quantity per liver within the completely different teams. ****p < 0.0001. D The liver-to-body weight ratio within the completely different remedy teams. **p < 0.01. E The variety of HCC nodules per liver in every group. *p < 0.05, ****p < 0.0001. F The tumor incidence in every group. G Pictures of H&E-stained liver sections exhibiting the histological construction and inflammatory response within the indicated teams. Black asterisks characterize accumulation of inflammatory cells. (H) Physique weight curves. The rats had been weighed each different week (n = 5 per group). ****p < 0.0001 in comparison with saline group. (I) Serological evaluation of creatine kinase (CK) was carried out after the rats had been sacrificed. Knowledge are introduced as imply ± SD. ns, not statistically vital
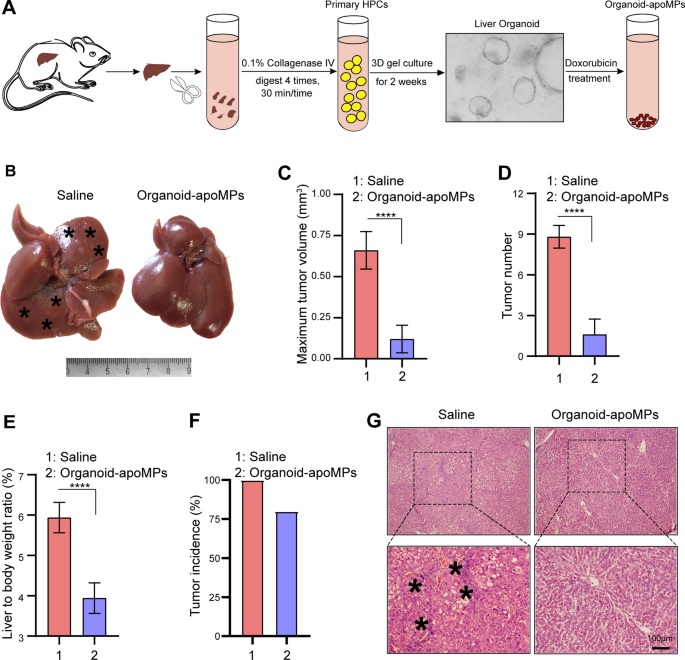
With a view to additional verify the position of apoHPC-MPs on hepatocarcinogenesis, we remoted major HPCs from rats by way of collagen IV digestion and fashioned liver organoids. The organoids had been handled with 100 µg/ml of doxorubicin, then organoid-apoMPs had been produced (Fig. 2A). With a view to take a look at the therapeutic efficacy of organoid-apoMPs, the rat major HCC mannequin was established and the animals had been intrasplenically handled with organoid-apoMPs or saline. Organoid-apoMPs (40 µg) had been administered to rats after 6 weeks of DEN remedy to induce hepatocarcinogenesis. Rats had been handled with MPs or saline twice per week for 7 weeks. On week 13 of DEN remedy, the rats had been sacrificed and the tumor development was evaluated. As proven in Fig. 2B, virtually no tumor nodes had been noticed within the organoid-apoMPs remedy group. In distinction, a number of tumor nodes had been noticed within the saline management group. Therapy with organoid-apoMPs additionally decreased the utmost tumor quantity (Fig. 2C), tumor quantity (Fig. 2D), liver-to-body weight ratio (Fig. 2E) and tumor incidence (Fig. 2F). In contrast with livers from the saline group, livers from the organoid-apoMPs group confirmed a decreased inflammatory response and a transparent tissue construction, as revealed by H&E staining (Fig. 2G). Due to this fact, major HPC-derived apoMPs effectively inhibit hepatocarcinogenesis.
MPs derived from apoptotic major HPCs stop hepatocarcinogenesis. A Experimental define for producing organoid-apoMPs. Rats had been handled with DEN for six weeks. Livers had been eliminated, minimize into small items, and digested with 0.1% collagenase IV. Major HPCs had been then remoted and cultured to type organoids. 100 µg/ml of doxorubicin was used to deal with the organoids and organoid-apoMPs had been then remoted. B After 6 weeks of DEN remedy, Sprague Dawley rats had been intrasplenically injected with 40 µg of organoid-apoMPs in 200 µl saline or 200 µl of clean saline. Injections got twice each week for 7 weeks. DEN remedy was additionally continued throughout this time. After 13 weeks, the rats had been sacrificed to watch the event of HCC. Consultant pictures of livers are proven. Typical tumor nodes are indicated by the asterisks. C The utmost tumor quantity per liver within the two teams. Knowledge are introduced as imply ± SD. ****p < 0.0001. D The variety of HCC nodules per liver. Knowledge are introduced as imply ± SD. ****p < 0.0001. E The liver-to-body weight ratio. Knowledge are introduced as imply ± SD. ****p < 0.0001. F The tumor incidence within the two teams. G Pictures of H&E-stained liver sections, exhibiting the histological construction and inflammatory response of the liver within the indicated teams. Black asterisks characterize accumulation of inflammatory cells
HPCs take up extra apoHPC-MPs than apoLTC-MPs and apoHep-MPs
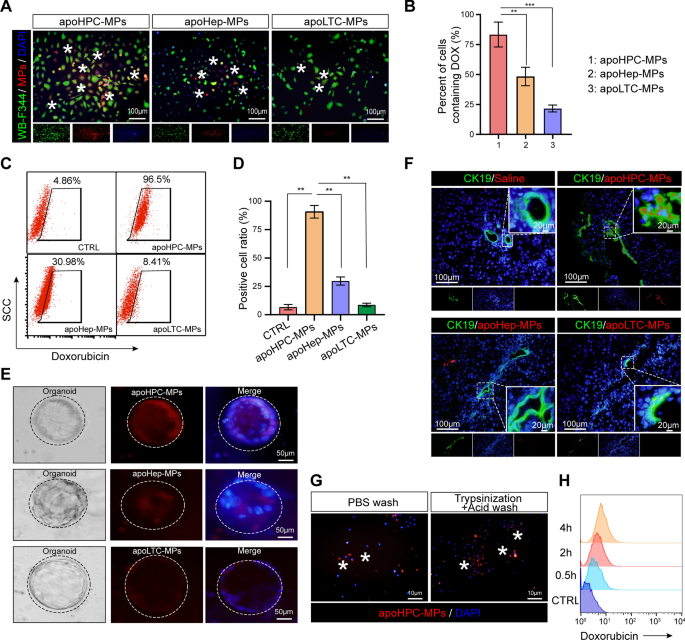
The above outcomes present that apoHPC-MPs stop hepatocarcinogenesis extra effectively than apoLTC-MPs and apoHep-MPs. To elucidate the underlying mechanism, we first requested whether or not HPCs preferntially take up apoHPC-MPs in comparison with apoLTC-MPs and apoHep-MPs. Due to this fact, doxorubicin (100 µg/ml) was added to cultured WB-F344 cells (1 × 107), RH35 cells (1 × 107) or BRL cells (1 × 107). After 12 h, the launched apoHPC-MPs, apoLTC-MPs and apoHep-MPs had been remoted from the tradition medium by centrifugation. The identical numbers of apoHPC-MPs (1 × 105), apoLTC-MPs (1 × 105) and apoHep-MPs (1 × 105) had been incubated with WB-F344 cells (5 × 103) for 30 min. Fascinating, solely the apoHPC-MPs, and never the apoLTC-MPs and apoHep-MPs, had been effectively taken up by WB-F344 cells (Fig. 3A). Round 75% of WB-F344 cells took up apoHPC-MPs (Fig. 3B). Moreover, the precise focusing on of apoHPC-MPs, apoLTC-MPs and apoHep-MPs was quantitatively decided by circulate cytometry evaluation. Equal numbers of apoHPC-MPs (1 × 106), apoLTC-MPs (1 × 106) and apoHep-MPs (1 × 106) had been incubated with WB-F344 cells (3 × 105) for 30 min. The outcomes indicated that 96% of WB-F344 cells had been constructive for apoHPC-MPs, whereas solely about 8% of WB-F344 cells had been constructive for apoLTC-MPs, and about 30% of WB-F344 cells had been constructive apoHep-MPs (Fig. 3C, D). This means that WB-F344 cells selectively take up apoHPC-MPs, relatively than apoLTC-MPs or apoHep-MPs. Constantly, RH35 cells confirmed a powerful choice for apoLTC-MPs, relatively than apoHPC-MPs and apoHep-MPs (Extra file 2: Fig. S5A). Primarily based on circulate cytometry evaluation, about 97% of RH35 cells had been constructive for apoLTC-MPs, whereas solely about 12% of RH35 cells took up apoHep-MPs and about 38% of RH35 cells took up apoHPC-MPs (Extra file 2: Fig. S5B). BRL cells additionally indicated a constant sample of uptake, with a powerful choice for apoHep-MPs, relatively than apoHPC-MPs and apoLTC-MPs (Extra file 2: Fig. S5C, D). These outcomes point out that cells can effectively take up parental cell-derived MPs. Thus, apoHPC-MPs effectively goal WB-F344 cells.
HPCs take up extra apoHPC-MPs than apoLTC-MPs or apoHep-MPs. A, B WB-F344 cell (5 × 103) had been labeled with GFP (inexperienced fluorescence), incubated for 30 min with 1 × 105 apoHPC-MPs, apoLTC-MPs or apoHep-MPs containing DOX (crimson fluorescence). The uptake of MPs into WB-F344 cells was noticed by fluorescence microscopy. Consultant pictures are proven in A. The proportion of WB-F344 cells with crimson fluorescence (indicating uptake of MPs) was calculated in every group. The mixed information from three experiments are proven in B. Knowledge are introduced as imply ± SD. **p < 0.01, ***p < 0.001. C, D 1 × 106 ApoHPC-MPs, apoLTC-MPs and apoHep-MPs containing doxorubicin (crimson fluorescence) had been incubated with WB-F344 cells (3 × 105) for 30 min. The proportion of WB-F344 cells containing crimson fluorescence was measured by circulate cytometry. Consultant circulate cytometry information are proven in C. The mixed information from three experiments are proven in (D). Knowledge are introduced as imply ± SD. **p < 0.01. E Liver organoids had been cultured in vitro. 1 × 104 apoHPC-MPs, apoLTC-MPs or apoHep-MPs had been incubated with liver organoids for 30 min. The uptake of MPs into organoids was noticed by fluorescence microscopy. Consultant pictures are proven. Nuclei had been stained with DAPI (blue). F 1 × 107 apoHPC-MPs, apoHep-MPs or apoLTC-MPs had been intrasplenically injected into DEN-treated rats. 30 min after injection, liver slices had been acquired for fluorescence detection. HPCs had been acknowledged by antibodies in opposition to CK19 (inexperienced). Nuclei had been stained with DAPI (blue). HPCs effectively took up crimson fluorescent apoHPC-MPs. G WB-F344 cells (3 × 105) had been incubated with apoHPC-MPs (1 × 105) for 30 min. Then WB-F344 cells had been handled with 0.2 × trypsin/EDTA buffer for 1 min after which washed with citric acid buffer a number of instances to take away all non-internalized apoHPC-MPs sure to the floor of WB-F344 cells. Nuclei had been stained with DAPI (blue). H 3 × 105 WB-F344 cells had been incubated with 2 × 105 apoHPC-MPs for 0.5, 2, and 4 h. After washing, the samples had been subjected to circulate cytometric evaluation. A consultant picture is proven right here
To additional take a look at the focusing on of apoMPs to HPCs, we remoted major HPCs from rat liver and generated organoids. Equal numbers of apoHPC-MPs (1 × 104), apoLTC-MPs (1 × 104) and apoHep-MPs (1 × 104) had been incubated with major HPCs for 30 min. Major HPCs effectively took up apoHPC-MPs, relatively than apoHep-MPs and apoLTC-MPs, as evidenced by fluorescence microscopy (Fig. 3E). As a result of all these findings had been primarily based on cultured HPCs, we needed to additional make clear whether or not apoMPs have the identical capability to focus on HPCs in vivo. For this experiment, 1 × 107 of apoHPC-MPs, apoHep-MPs and apoLTC-MPs had been intrasplenically injected into DEN-treated rats and liver slices had been acquired for fluorescence detection 30 min after injection. The expression of the HPC marker CK19 in liver sections was examined [24,25,26]. As noticed in cell tradition, accumulation of crimson fluorescent apoHPC-MPs, relatively than apoHep-MPs and apoLTC-MPs, was present in CK19-positive HPCs (Fig. 3F). This demonstrates that HPCs can selectively take up HPC-derived apoHPC-MPs in vivo.
We additionally examined the interplay of HPCs and apoHPC-MPs. WB-F344 cells (3 × 105) had been incubated with apoHPC-MPs (1 × 105) for 30 min, then handled with trypsin and washed a number of instances with citric acid to take away the surface-bound MPs. Fluorescence imaging revealed that apoHPC-MPs weren’t cleared from WB-F344 cells after trypsinization and acid washing (Fig. 3G). This means that apoHPC-MPs had been internalized into WB-F344 cells, relatively than being connected to the floor of the WB-F344 cells. We additional studied the dynamics of apoHPC-MPs internalization into WB-F344 cells. For this experiment, 3 × 105 WB-F344 cells had been incubated with 2 × 105 apoHPC-MPs. The internalization of apoHPC-MPs was detected after 0.5 h and elevated markedly with time (Fig. 3H), suggesting that the uptake of apoHPC-MPs by HPCs is time-dependent.
ApoHPC-MPs are cytotoxic to HPCs
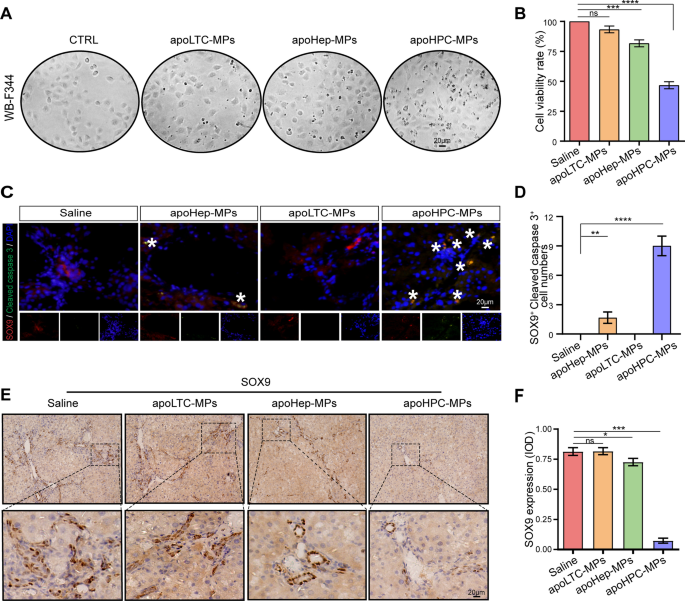
Subsequent, we requested if apoHPC-MPs are cytotoxic to HPCs after they’re internalized. ApoHPC-MPs (20 µg), apoLTC-MPs (20 µg) or apoHep-MPs (20 µg) had been added to cultured WB-F344 cells (5 × 103) for twenty-four h. In contrast with apoHep-MPs and apoLTC-MPs, apoHPC-MPs induced extra killing of WB-F344 cells (Fig. 4A). Furthermore, the proliferation of WB-F344 cells was effectively inhibited by remedy with apoHPC-MPs (Fig. 4B). To check this end result in vivo, 40 µg of apoHPC-MPs, apoHep-MPs or apoLTC-MPs had been intrasplenically injected into rats that beforehand obtained DEN remedy for six weeks. After 24 h, liver slices had been acquired for fluorescence detection. HPCs had been labeled with an antibody in opposition to SOX9 [26, 27] and apoptotic cells had been detected with an antibody in opposition to cleaved caspase 3. The best variety of apoptotic HPCs (constructive for each SOX9 and cleaved caspase 3) was seen within the apoHPC-MPs remedy group (Fig. 4C, D). Within the apoHep-MPs group, a small variety of HPCs had been constructive for cleaved caspase 3 staining, whereas saline and apoLTC-MPs didn’t induce apoptosis of HPCs (Fig. 4C, D). To corroborate these observations, we additionally evaluated the expression of the HPC marker SOX9 and CK7 in liver sections by IHC [23, 24]. ApoLTC-MPs and the management group confirmed intense and diffuse constructive SOX9 and CK7 staining, whereas apoHep-MPs brought about a small lower within the SOX9 and CK7 sign and apoHPC-MPs brought about a big lower in SOX9 and CK7 staining (Fig. 4E, F and Extra file 2: Fig. S6). Due to this fact, after injection of apoMPs into rats, the variety of HPCs was vastly decreased by apoHPC-MPs, partly decreased by apoHep-MPs, and unaffected by apoLTC-MPs. Because of all these experiments, we conclude that HPCs can selectively take up cytotoxic apoHPC-MPs, inhibiting the proliferation of hepatoma-initiating cells.
ApoHPC-MPs are cytotoxic to HPCs. A The identical quantity (20 µg) of apoLTC-MPs, apoHep-MPs or apoHPC-MPs was incubated with cultured WB-F344 cells (5 × 103) for twenty-four h. The killing of WB-F344 cells was noticed by microscopy. B WB-F344 cells had been handled with 20 µg of apoHPC-MPs, apoHep-MPs or apoLTC-MPs for 1 h. The cells had been washed and cultured in contemporary medium for twenty-four h. Cell proliferation was detected by CCK8 assay. Knowledge had been collected from three unbiased experiments. Knowledge are introduced as imply ± SD. ***p < 0.001, ****p < 0.0001. C 40 µg of apoHPC-MPs, apoHep-MPs or apoLTC-MPs had been intrasplenically injected into rats, which had been handled with DEN for the earlier 6 weeks. 24 h after injection, liver slices had been acquired for fluorescence detection. HPCs had been stained with antibodies in opposition to SOX9 (crimson) and cleaved caspase 3 (inexperienced) for fluorescence microscopy evaluation. Nuclei had been stained with DAPI (blue). Consultant footage of staining are proven. White asterisks present the SOX9-positive HPCs that are additionally constructive for cleaved caspase 3. D Cells constructive for each SOX9 and cleaved caspase 3 had been counted in every area of view in C; the mixed information from three experiments are proven. **p < 0.01, ****p < 0.0001 E 40 µg of apoHPC-MPs, apoHep-MPs or apoLTC-MPs had been intrasplenically injected into rats, which had been handled with DEN for the earlier 6 weeks. Injections had been administered twice each week for 4 weeks. Rats had been then sacrificed, and liver tissue sections had been acquired for IHC assay. The HPCs had been stained with an antibody in opposition to SOX9. Consultant pictures are proven. Within the apoHPC-MP-treated liver, SOX9 staining is faint and really scarce. The apoHep-MP-treated liver exhibits a reasonable degree of SOX9 staining. The opposite livers present sturdy and diffuse staining. F Laptop-assisted IOD analysis of SOX9 staining in E. Knowledge had been collected from three unbiased experiments. Knowledge are introduced as imply ± SD. *p < 0.05, ***p < 0.001
MPs from non-apoptotic HPCs don’t have any have an effect on hepatocarcinogenesis
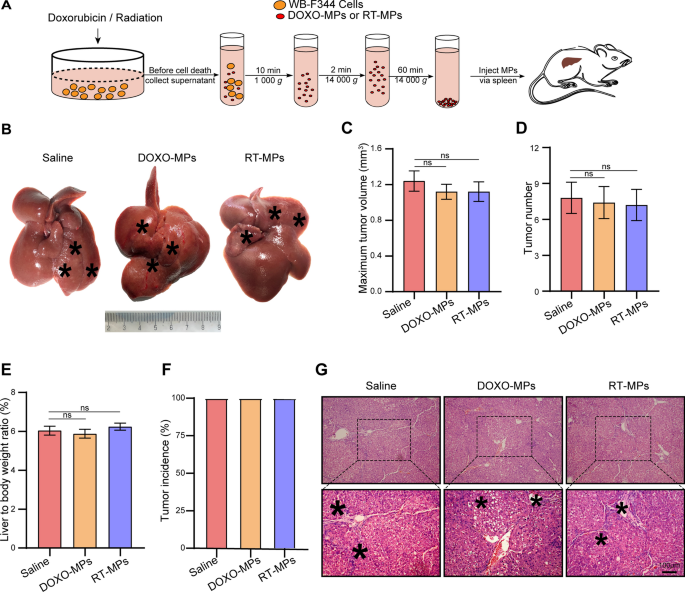
To additional make clear the position of the demise sign, we generated MPs from non-apoptotic HPCs. WB-F344 cells had been handled with 100 µg/ml of DOX, or irradiated with 20 Gy, after which MPs with out demise sign (DOXO-MPs and RT-MPs) had been collected from the cells earlier than they underwent apoptosis. 40 µg of DOXO-MPs and RT-MPs had been injected into rats, which had been handled with DEN for six weeks (Fig. 5A). MPs or saline had been injected twice each week for 7 weeks. The rats had been then sacrificed to watch the tumor development. As proven in Fig. 5B, in contrast with the saline management group, remedy with DOXO-MPs and RT-MPs had no impact on hepatocarcinogenesis. This end result was additionally confirmed by the utmost tumor quantity (Fig. 5C), tumor quantity (Fig. 5D), liver-to-body weight ratio (Fig. 5E) and tumor incidence (Fig. 5F). H&E staining confirmed that the inflammatory response was related within the livers of every group (Fig. 5G). Due to this fact, MPs derived from non-apoptotic HPCs had no impact on hepatocarcinogenesis.
HPC-derived MPs with out demise alerts don’t have any impact on hepatocarcinogenesis. A Experimental define for producing MPs with out apoptotic alerts (DOXO-MPs and RT-MPs). WB-F344 cells had been handled with 100 µg/ml doxorubicin or 20 Gy radiation. DOXO-MPs and RT-MPs had been remoted earlier than the cells underwent apoptosis. B–F Sprague Dawley rats, which had been handled with DEN for six weeks, had been intrasplenically injected with 40 µg of DOXO-MPs and RT-MPs. Injections had been administered twice each week for 7 weeks. DEN remedy was additionally continued throughout this time. After 13 weeks, the rats had been sacrificed to watch the event of hepatocellular carcinoma (HCC). Consultant pictures of rat livers are proven. Typical tumor nodes are marked by the asterisks (B). The utmost tumor quantity per liver within the indicated teams (C). The variety of HCC nodules per liver (D). The liver-to-body weight ratio (E). The tumor incidence within the completely different remedy teams (F). G Pictures of H&E-stained liver sections exhibiting the histological construction and inflammatory response after the indicated therapies. Black asterisks point out accumulation of inflammatory cells
As a result of apoHPC-MPs had been produced by treating HPCs with doxorubicin, the launched apoHPC-MPs contained doxorubicin along with the demise sign (Extra file 2: Fig. S1C). To exclude any impact of the therapeutic drug doxorubicin on killing HPCs by apoHPC-MPs within the rat major HCC mannequin, the quantity of doxorubicin in apoHPC-MPs was detected by liquid chromatography-mass spectrometry (LC-MS) evaluation (Extra file 2: Fig. S7A). Free doxorubicin (~ 10 µg, which was the identical quantity of doxorubicin encapsulated in apoHPC-MPs for injection of every rat) was intrasplenically injected into rats, which had been handled with DEN for six weeks. Injections had been carried out twice each week for 7 weeks. The rats had been then sacrificed for tumor examination. In contrast with the saline management group, the free doxorubicin had no vital impact on hepatocarcinogenesis (Extra file 2: Fig. S7B). There was no vital distinction within the most tumor quantity (Extra file 2: Fig. S7C), tumor quantity (Extra file 2: Fig. S7D), liver-to-body weight ratio (Extra file 2: Fig. S7E) and tumor incidence (Extra file 2: Fig. S7F). H&E staining confirmed that the inflammatory response was related within the livers of every group (Extra file 2: Fig. S7G). Due to this fact, doxorubicin encapsulated in apoHPC-MPs had no impact on hepatocarcinogenesis.
HPC-derived MPs containing demise alerts inhibit hepatocarcinogenesis within the absence of doxorubicin
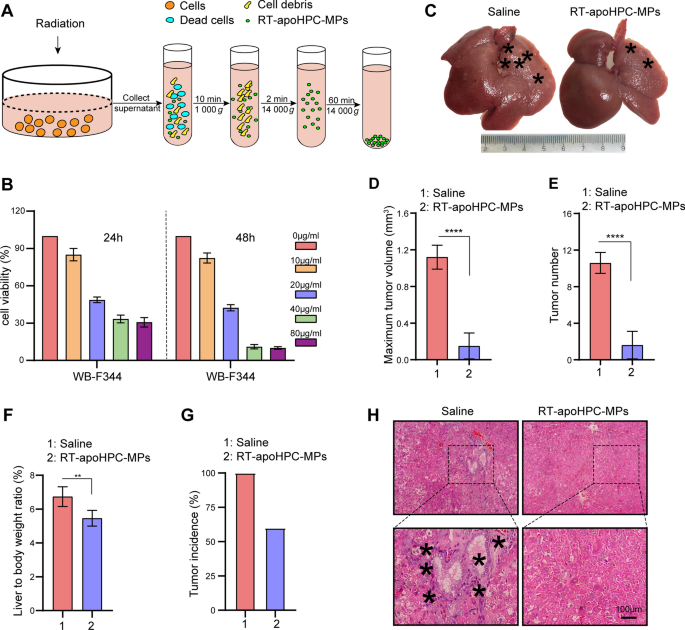
To additional make clear the position of the demise sign in MPs, and exclude the impact of the encapsulated chemotherapeutic drug, we needed to isolate MPs with out chemotherapeutic medication. To do that, we used a deadly dose of radiation to induce WB-F344 cell demise. WB-F344 cells had been handled with varied doses of radiation to determine the deadly dose. We noticed elevated cell killing with an elevated radiation dose till 20 Gy, which induced the utmost degree of cell demise (Extra file 2: Fig. S8A). The quantity of MPs differed in line with the next tradition time of cells after 20 Gy radiation: the maximal quantity of MPs was remoted after culturing WB-F344 cells for one more 72 h (Extra file 2: Fig. S8B). We selected these situations for subsequent research. The MPs had been designated as RT-apoHPC-MPs (Fig. 6A). To additional consider the therapeutic impact of RT-apoHPC-MPs on HPCs, we carried out cell toxicity research on the HPC cell line WB-F344. We discovered that RT-apoHPC-MPs effectively inhibited the expansion of WB-F344 cells in a dose- and time-dependent method (Fig. 6B and Extra file 2: Fig. S8C). We then investigated the therapeutic impact of RT-apoHPC-MPs within the rat major HCC mannequin. RT-apoHPC-MPs (40 µg) had been intrasplenically injected into rats, which had been handled with DEN for six weeks. Injections had been carried out twice each week for 7 weeks. The rats had been then sacrificed for tumor examination. As proven in Fig. 6C, we noticed decreased tumor development upon remedy with RT-apoHPC-MPs, superior to that of the management remedy. This end result was supported by evaluation of most tumor quantity (Fig. 6D), tumor quantity (Fig. 6E), liver-to-body weight ratio (Fig. 6F) and tumor incidence (Fig. 6G). H&E staining confirmed a decreased inflammatory response and clear tissue construction within the livers of rats handled with RT-apoHPC-MPs (Fig. 6H). Due to this fact, RT-apoHPC-MPs carrying a demise sign can effectively inhibit hepatocarcinogenesis within the absence of doxorubicin.
HPC-derived MPs carrying the demise sign inhibit hepatocarcinogenesis within the absence of doxorubicin. A Experimental define for producing RT-apoHPC-MPs. WB-F344 cells had been handled with 20 Gy of radiation and RT-apoHPC-MPs had been remoted after apoptosis of the cells. B WB-F344 cells had been handled with varied concentrations of RT-apoHPC-MPs for twenty-four or 48 h, and cell viability was detected utilizing the CCK-8 assay. C–G Rats had been handled with DEN for six weeks, then intrasplenically injected with RT-apoHPC-MPs (40 µg). Injections had been administered twice each week for 7 weeks. DEN was administered orally in parallel. After 13 weeks, the rats had been sacrificed to watch the event of HCC. Consultant pictures of rat livers are proven. Typical tumor nodes are marked by the asterisks (C). The utmost tumor quantity per liver within the two teams (D). The variety of HCC nodules per liver (E). The liver-to-body weight ratio within the two teams (F). The tumor incidence within the two teams (G). Knowledge are introduced as imply ± SD. **p < 0.01, ****p < 0.0001. H Pictures of H&E-stained liver sections exhibiting the histological construction and inflammatory response of liver within the two teams. Black asterisks point out accumulation of inflammatory cells
Quantitative proteomic evaluation of extracellular microparticles reveals enrichment of death-related proteins in apoHPC-MPs
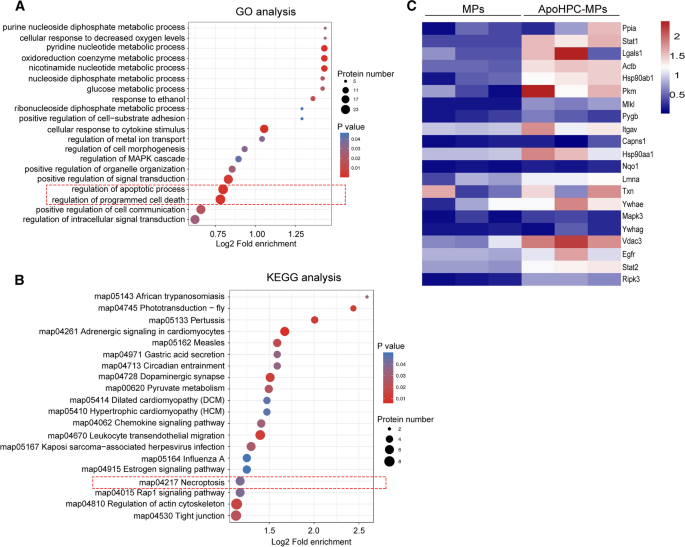
To additional examine the demise sign encapsulated by apoHPC-MPs, quantitative proteomics evaluation was carried out on apoHPC-MPs from apoptotic WB-F344 cells and MPs from non-apoptotic WB-F344 cells by MS/MS. Three unbiased experiments reliably recognized 523, 344 and 652 proteins from MPs, and 310, 769 and 978 proteins from apoHPC-MPs. Of those, 278 proteins had been quantitatively recognized in all replicates (Extra file 1: Desk S1). The edge for vital up-regulation was set as fold change > 1.5 and the brink for vital down-regulation was set as lower than 1/1.5. Underneath these standards, there have been 116 proteins considerably upregulated in apoHPC-MPs as in comparison with MPs (Extra file 1: Desk S1). Gene ontology (GO) enrichment evaluation indicated that the 116 up-regulated proteins had been enriched in organic processes associated to regulation of apoptotic course of and regulation of programmed cell demise (Fig. 7A). Additional, Kyoto Encyclopedia of Genes and Genomes (KEGG) Evaluation confirmed that the 116 up-regulated proteins had been additionally associated to the necroptosis pathway (Fig. 7B). Furthermore, primarily based on 116 up-regulated proteins, gene encoding death-related proteins are current at greater ranges within the apoHPC-MPs than in MPs (Fig. 7C). Collectively, these outcomes point out that apoHPC-MPs inhibit hepatocarcinogenesis by transmitting a demise sign to hepatoma-initiating cells, which ends up in the demise of hepatoma-initiating cells.
Quantitative proteomic evaluation of extracellular microparticles reveals enrichment of demise associated proteins in apoHPC-MPs. A Differentially expressed (up-regulated) proteins between apoHPC-MPs and MPs had been labeled by GO annotation primarily based on the next class: organic course of. The bubble chart exhibits the highest 20 classes with probably the most vital enrichment. The vertical axis exhibits the perform classification. The x-axis signifies Log2 of the ratio of the variety of differential proteins within the corresponding perform classification to the variety of whole proteins recognized. The colours of the factors characterize the P values of enrichment significance. The sizes of the dots characterize the numbers of differential proteins within the corresponding perform classification. B Annotation of differentially expressed (up-regulated) proteins between apoHPC-MPs and MPs primarily based on the KEGG pathway database. The bubble chart exhibits the highest 20 classes with probably the most vital enrichment. The vertical axis exhibits the practical pathway. The x-axis signifies Log2 of the ratio of the variety of differential proteins within the corresponding pathway to the variety of whole proteins recognized. The colours of the factors characterize the P values of enrichment significance. The sizes of the dots characterize the numbers of differential proteins within the corresponding pathway. C Primarily based on the MS outcomes, warmth map of the up-regulated gene encoding death-related proteins in apoHPC-MPs in comparison with MPs
[ad_2]