[ad_1]
A French hospital put in a bioprinter to fabricate organic tissues for transplantation in people. This quantum leap in bioprinting is feasible because of Poietis, an area biotech firm specializing within the design of regenerative drugs therapies and proprietary 4D laser bioprinting know-how. Because the recipient of Poietis’ Subsequent-Era Bioprinting (NGB) robotic system, the Hôpital de la Conception, which is a part of the Marseille Public College Hospital System (AP-HM), will begin utilizing the gadget as a part of a scientific trial for grafting bioprinted pores and skin tissue utilizing sufferers’ personal cells.
Put in on the Hôpital de la Conception’s Superior Remedy Medicinal Product (ATMP) manufacturing space, the gadget represents an actual technological revolution for the healthcare sector. The truth is, it’s the results of a collaboration between Poietis and the AP-HM’s Cell Tradition and Remedy Laboratory (LCTC) that started in early 2020 to beat the challenges of presently obtainable bioprinting processes.
In line with Poietis, the present manufacturing strategies of organic tissues are usually not ok to be adopted by the medical neighborhood, regardless that a number of extraordinarily promising advances paved the way in which for brand spanking new therapeutic methods in tissue restore and substitution. Specialists at AP-HM and Poietis think about that the “manufacturing processes have to be standardized,” and coverings must be extra inexpensive.
Up till now, varied bioprinting know-how platforms have been developed to fulfill these challenges. Nonetheless, none of them has proved suitable with the Good Manufacturing Practices rules related to manufacturing superior remedy medicinal merchandise (ATMPs). So, no bioprinted organic tissue has but been implanted in a affected person.
Commenting on the brand new on-site bioprinting know-how, Poietis founder and president Fabien Guillemot stated, “bioprinting is coming into a brand new period. The event of the primary bioprinting platform that complies with the Good Manufacturing Practices (GMP) of ATMPS is a breakthrough innovation within the area of regenerative drugs because it removes one of many final boundaries earlier than grafting bioprinted tissues into sufferers.”
The NGB bioprinting platform, developed by Poietis and not too long ago put in within the AP-HM hospital, meets these challenges because of quite a few technological advances, together with the mixing of laser-assisted bioprinting and 3D extrusion biomaterial printing applied sciences in a very aseptic setting. Furthermore, the robotization of the platform and the automation of the processes enable the manufacturing of purposeful tissues with a excessive degree of reproducibility and suppleness.
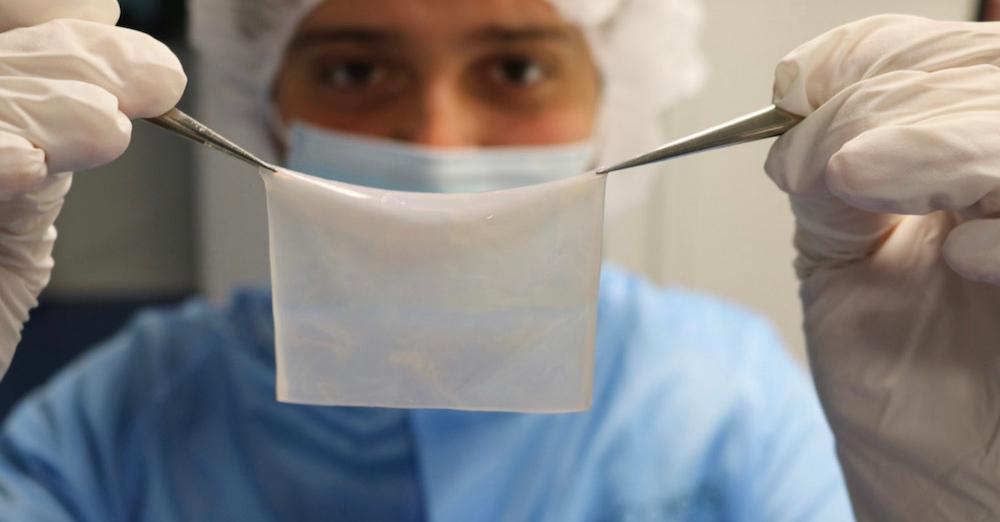
As if that weren’t sufficient, the collaboration between Poietis and the French hospital resulted in a thousandfold elevated printing pace, which lets customers make 40 sq. centimeters (6.2 sq. inches) of pores and skin substitutes in a couple of hours. As a reference, British specialists have estimated the typical floor space of the pores and skin in grownup girls to be roughly 16,000 sq. centimeters (2,480 sq. inches) and 18,000 sq. centimeters (2,790 sq. inches) in grownup males.
Medical doctors will be capable of bioprint a big amount of pores and skin on the hospital because of a easy pattern taken very simply from the affected person. This can be a therapeutic innovation that AP-HM plastic surgeon, Maxime Abellan-Lopez, says he has been ready for. It “gives us the capability to reconstruct the pores and skin of sufferers requiring a graft, akin to after a burn or critical trauma, with minimal hostile results.”
The workforce on the hospital will bioprint a proprietary tissue mannequin engineered by Poietis often called Poieskin. Described as a “human full-thickness pores and skin mannequin,” Poieskin consists of a dermal compartment composed of major human fibroblasts embedded in a collagen I matrix overlaid by a stratified dermis (the outermost layer of the pores and skin) and derived from major human keratinocytes–the first kind of cell discovered within the dermis. As for the biofabrication course of, it begins with dermis bioprinting and dermis maturation, adopted by keratinocytes bioprinting, epidermal proliferation, and stratification on the air-liquid interface.
A mature dermis features as a barrier that protects the physique from dehydration or trauma. Recreating it utilizing a bioprinting functionality like Poietis may enhance the restore of burn accidents. Sometimes, burn therapy, particularly within the case of third- and fourth-degree burns, require specialised medical care involving the surgical elimination of the injured pores and skin and reconstruction of the burn damage utilizing pores and skin substitutes, like grafts. Ever since bioprinting has been steered for treating burn wounds, the potential purposes for the know-how have been very promising. Admittedly, the scientific trial translation should meet the required technological and regulatory challenges earlier than it turns into obtainable to sufferers.
Nonetheless, Poietis and the AP-HM are transferring quick. Due to the most recent suggestions from the French Company for the Security of Well being Merchandise, the LCTC will finalize the validation of the Poieskin pores and skin substitute manufacturing course of earlier than beginning a scientific trial subsequent yr, which can be carried out with the help of the AP-HM’s Plastic and Reconstructive Surgical procedure Division and the Interregional Middle for extreme burns.
Though the medical middle is concentrated on grafting human tissue utilizing a bioprinter, the final word aim is to bioprint extra advanced tissues on the level of care, as near the affected person as potential. Like many medical docs, the workforce at AP-HM desires about equipping hospitals with bioprinters, the place at-risk sufferers can have entry to the factitious creation of human pores and skin, tissue, and even sometime, inside organs. Though this appears distant into the long run, Poietis is readying the idea for what’s to come back.
“Sooner or later, we are able to envision all main hospitals being geared up with bioprinters. This jogs my memory of the success of surgical robotics: a disruptive innovation that has turn into important in every day observe,” described LCTC pharmacist-biologist Jérémy Magalon.
For now, this set up is a big milestone that can set off the commercialization of NGB-C bioprinters to cell remedy and translational analysis facilities in France and ultimately Europe and the US. It is going to additionally assist Poietis speed up the event of its portfolio of implantable bioprinted tissues for wound therapeutic within the osteoarticular area and for treating neurodegenerative illnesses.
[ad_2]