[ad_1]
Design, synthesis and characterization of M@P-PDR
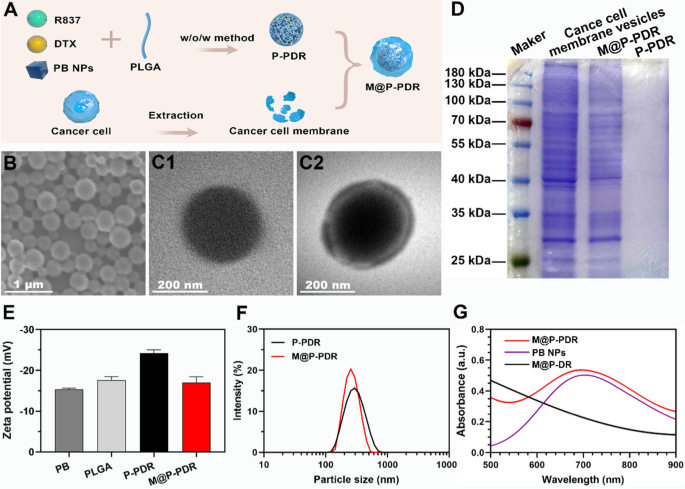
M@P-PDR nanospheres, distinctive “Nano-targeted cells”, have been constructed by coating P-PDR nanospheres with most cancers cell membranes with homologous focusing on functionality (Fig. 1A). Initially, P-PDR nanospheres have been ready by a typical double- emulsion technique (water/oil/water), which encapsulates hydrophobic medicine within the PLGA shell layer and hydrophilic medicine within the core of PLGA nanospheres [41,42,43]. PB nanoparticles are hydrophilic medicine and might be dispersed effectively in aqueous answer (Further file 1: Fig. S1). Each R837 and DTX are lipid-soluble medicine. Due to this fact, PB is encapsulated within the core of PLGA nanospheres, whereas R837 and DTX are current within the shell. Coextrusion was probably the most adopted technique to coat nanoparticles with membrane. As a result of fluidity of the cell membrane, the mechanical pressure exerted by the extrusion course of promote the nanoparticles to be encapsulated by the phospholipid layer [33, 44]. Nonetheless, this method is a tedious and time-consuming course of. Happily, sonication is an efficient various to extrusion and used to organize M@P-PDR nanospheres on this examine [38]. Cell membranes could be destroyed by ultrasonic waves and the fragments might be self-assembled across the nanospheres. This one-step fabrication is facile method. It has been discovered that repulsion between negatively charged nanoparticle-core and cell membrane-shell permits profitable membrane coating with a “right-side-out” membrane topological method orientation [45, 46]. The SEM picture indicated that M@P-PDR displayed a uniform and spherical morphology (Fig. 1B). M@P-PDR “Nano-targeted cells” confirmed a extra apparent coating than P-PDR (Fig. 1C), which might be ascribed to protection by most cancers cell membranes. Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) was used to additional analyze the protein composition of those M@P-PDR “Nano-targeted cells”, and the outcomes confirmed that M@P-PDR nanospheres had nearly the identical protein composition as the unique 4T1 cell membrane (Fig. 1D), which additional demonstrated the success of cell membrane coating. Excitingly, the P-PDR nanospheres are negatively charged (Fig. 1E), which ensures the orientation of the cell membrane through the coating course of, thereby sustaining the organic operate of the cell membrane [46]. The zeta potentials of M@P-PDR nanospheres was − 17.0 ± 1.40 mV, which may probably lengthen blood circulation and profit different purposes within the organic milieu [47].
Characterization of the M@P-PDR nanospheres. A Schematic illustration of the artificial course of for M@P-PDR nanospheres. B SEM picture of M@P-PDR. C1 TEM picture of P-PDR; C2 TEM picture of M@P-PDR. D SDS-PAGE protein evaluation outcomes of most cancers cell membrane vesicles, M@P-PDR and P-PDR. E Zeta-potential of PB, PLGA, P-PDR and M@P-PDR nanospheres (n = 3). F DLS outcomes of P-PDR and M@P-PDR nanospheres. G UV–Vis-NIR spectra of PB NPs, M@P-DR and M@P-PDR suspensions
Dynamic gentle scattering (DLS) confirmed that the typical hydrodynamic diameter of the nanospheres barely elevated from 297 nm to 326.4 nm after cell membrane coating (Fig. 1F). We subsequent analyzed the PB nanoparticles, medicine (R837 and DTX), PLGA, P-P, P-PDR and M@P-PDR by FTIR. As proven in Further file 1: Fig. S2, PB, P-P, P-PDR and M@P-PDR have attribute absorption peaks at 2090 cm−1, which is exclusive to PB nanoparticles, demonstrating that PB has been efficiently encapsulated in PLGA [43]. M@P-PDR solely confirmed absorption peaks which additionally appeared in PB nanoparticles, PLGA, DTX, R837 and P-PDR, however no different new attribute absorption peaks have been noticed, additional indicating that the binding between the parts of the nanoparticles is bodily reasonably than chemical. In comparison with the UV–Vis spectrum of M@P-DR, the spectrum of the M@P-PDR suspension introduced a attribute absorption band of PB at 700 nm (Fig. 1G), indicating the profitable loading of PB nanoparticles in M@P-PDR. The loading efficacies of PB nanoparticles, R837 and DTX have been calculated to be 37.28%, 78.57% and 69.67%, respectively, in line with the usual curves (Further file 1: Fig. S3A, B) and liquid–mass spectrometry evaluation (Further file 1: Fig. S3C, D). Such excessive loading capacities for these medicine demonstrated that PLGA nanospheres have nice potential as promising nanocarriers for drug supply, which has additionally been reported by many earlier research [48, 49].
In vitro photothermal efficiency and PA/MR bimodal imaging of M@P-PDR
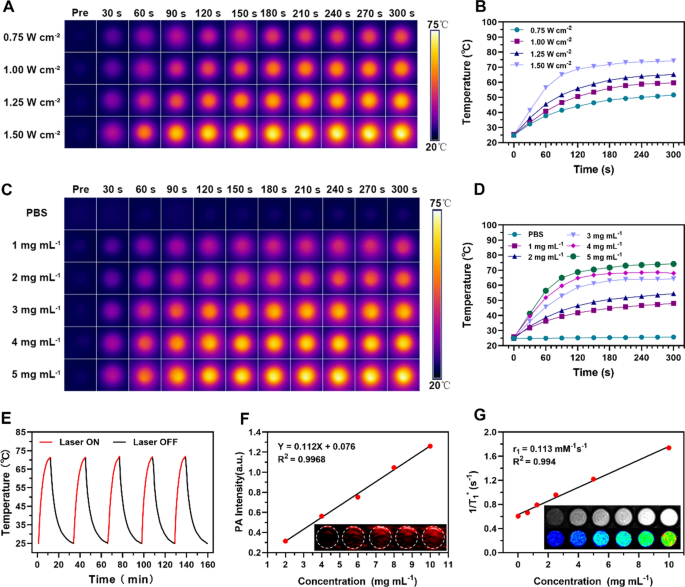
The distinctive absorbance of PB nanoparticles within the NIR area indicated the potential for M@P-PDR to spice up photothermal therapeutics [50]. Due to this fact, the in vitro photothermal efficiency of M@P-PDR was systemically studied. The photothermal conversion of M@P-PDR nanospheres was evaluated at totally different laser energy densities (0.75, 1.00, 1.25 and 1.50 W cm˗2) and totally different M@P-PDR concentrations (1, 2, 3, 4 and 5 mg mL˗1). Vital laser-power-dependent (Fig. 2A, B) and concentration-dependent (Fig. 2C, D) photothermal results have been noticed. Furthermore, wonderful photothermal heating/cooling-cycling stability was additionally demonstrated (Fig. 2E). On this floor, M@P-PDRs can be utilized as PTCAs for subsequent PTT.
A Infrared thermal photos of M@P-PDR at a focus of 5 mg mL−1 underneath 808 nm laser irradiation at totally different energy densities (0.75, 1.00, 1.25 and 1.50 W cm−2), and B the corresponding temperature–time curves of M@P-PDR at totally different energy densities. C Infrared thermal photos of M@P-PDR at totally different concentrations (0, 1, 2, 3, 4 and 5 mg mL−1) underneath 808 nm laser (1.5 W cm˗2, 5 min) irradiation, and D the corresponding photothermal temperature–time curves of M@P-PDRs at totally different concentrations. E Temperature change curves of M@P-PDR over 5 laser irradiation on/off cycles. F Linear relationship between PA intensities and M@P-PDR concentrations, and the corresponding in vitro PA photos (inset). G T1 rest price of M@P-PDR and the corresponding in vitro MR photos (inset)
The excessive sensitivity and excessive spatial decision of PA imaging facilitate the visualization of nanocarriers in vivo [51]. The multiwavelength PA sign spectrum of M@P-PDR nanospheres confirmed that 740 nm was the optimum wavelength for PA imaging (Further file 1: Fig. S4). As proven in Fig. 2F, the PA sign intensities of M@P-PDR suspensions elevated in a major concentration-dependent method. MR imaging efficiency was additionally investigated. As proven in Fig. 2G (inset), the brightness of the T1-weighted MR photos elevated with the focus of M@P-PDR nanospheres, and the pseudo-colored T1-mapping photos additionally confirmed the identical tendency. The comfort price (R1 worth) was calculated to be 0.113 mM−1 s−1 by measuring the relief time (Fig. 2G). With the improved PA/MR dual-modal imaging capability, the metabolic profiles of those M@P-PDR nanospheres at tumor websites might be visualized, offering steerage/monitoring for subsequent cocktail remedy.
Biocompatibility assay of M@P-PDR
As a prerequisite for any medical improvement, the biocompatibility of M@P-PDRs was investigated each in vitro and in vivo. First, the cytotoxicity of M@P-PDR and P-PDR nanospheres towards 4T1 cells was evaluated utilizing a typical CCK-8 assay. After 24 h of coincubation, each M@P-PDR and P-PDR nanospheres confirmed negligible toxicity to 4T1 cells when the PLGA focus was decrease than 400 μg mL−1 (Further file 1: Fig. S5). To additional examine the biocompatibility of M@P-PDR, the in vivo acute and comparatively long-term toxicity of M@P-PDR was evaluated in wholesome BALB/c mice. Routine blood exams and serum biochemical assays have been carried out on Day 1, 3, 7, 15 and 30 after the intravenous administration of M@P-PDR (Further file 1: Fig. S6A, B). In contrast with the reference vary of hematology information, all indicators of the handled mice and the management group remained at regular ranges. As well as, the main organs (coronary heart, liver, spleen, lung and kidney) have been collected for H&E staining (Further file 1: Fig. S6C), and negligible histomorphological or pathological adjustments have been noticed. All these outcomes strongly demonstrated the perfect excessive biocompatibility of M@P-PDR nanospheres as a multitasking therapeutic agent, offering nice potential for his or her additional medical translation.
In vitro homologous focusing on capability of M@P-PDR
The efficient intracellular uptake of M@P-PDR nanospheres is the important thing to their therapeutic efficacy. Functionalized by adhesion proteins of most cancers cells on the floor, most cancers cell biomimetic nanoplatforms are anticipated to exhibit particular homologous focusing on capability [42, 52, 53]. Western blotting outcomes confirmed the presence of those homologous binding adhesion molecules (EpCAM and galectin-3) on M@P-PDR (Further file 1: Fig. S7), which may obtain particular recognition and binding between supply cells particularly focusing on M@P-PDR and most cancers cells by a homologous binding mechanism. To analyze the specificity of M@P-PDRs to homologous 4T1 cells, the focusing on functionality of M@P-PDRs to 4T1 cells, MDA-MB-231 cells and SKBR3 cells was verified by CLSM and move cytometry. The outcomes confirmed that the 4T1 group had the very best uptake effectivity and fluorescence depth, demonstrating the precise binding capacity of M@P-PDRs to homologous 4T1 cells. (Further file 1: Fig. S8). Subsequent, the focusing on capability of those M@P-PDR “Nano-targeted cells” to 4T1 cells was evaluated utilizing CLSM. As proven in Fig. 3A, Further file 1: S9A, and B, 4T1 cells handled with M@P-PDR “Nano-targeted cells” exhibited stronger purple fluorescence than that of P-PDR nanospheres, indicating that the most cancers cell membrane-coating promoted the intracellular uptake of nanocarriers. Furthermore, the purple fluorescence enhanced with the extension of coincubation time. This phenomenon was additional confirmed by move cytometry quantitative analyses (Fig. 3B). As an example, after 2 h of incubation, the intracellular uptake price of the “Nano-targeted cells”-treated group reached 61.67%, whereas that of the P-PDR nanospheres-treated group was solely 12.54%.
A CLSM photos of 4T1 cells handled with M@P-PDR and P-PDR nanospheres for various occasions (0.5, 1, 2, 3 and 4 h), respectively (the blue signifies nucleus stained with DAPI, the purple signifies M@P-PDR or P-PDR nanospheres stained with DiI), and B the corresponding move cytometry quantitative analyses
The above outcomes indicated that the presence of most cancers cell membranes facilitated the intracellular uptake of nanocarriers thus exerting more practical therapeutic results.
In vitro therapeutic results
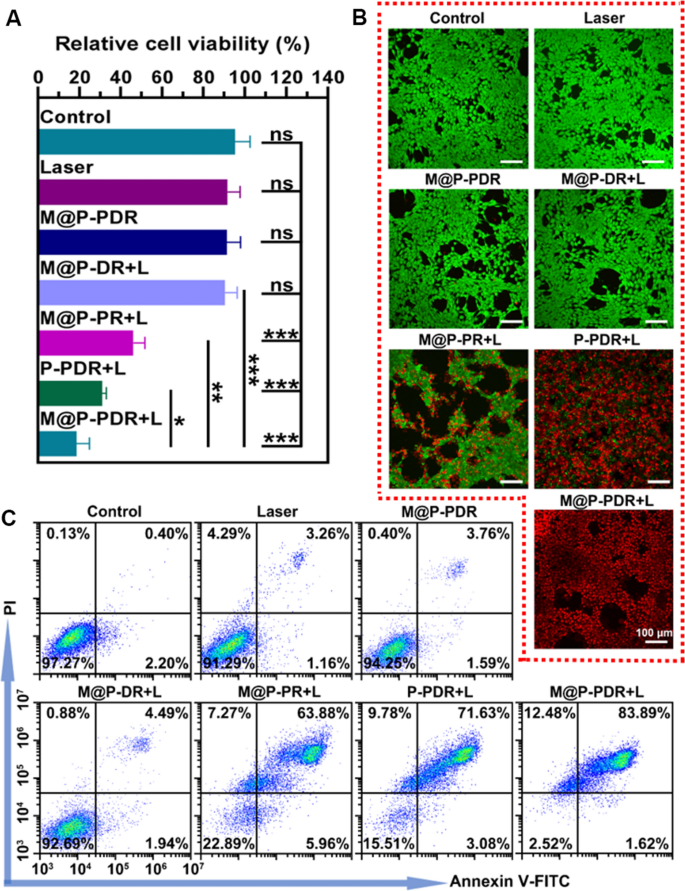
M@P-PDR nanospheres have been demonstrated to behave as PTCAs to transform gentle vitality into thermal vitality. The photothermal results mixed with a chemotherapeutic drug (DTX) of M@P-PDR nanospheres in opposition to 4T1 cells have been evaluated subsequent. In accordance with the outcomes of the CCK-8 assay (Fig. 4A), the cell viability in M@P-PR + L group was 46.14 ± 5.62%, exhibiting the excessive efficacy of PTT in opposition to tumor cells. The M@P-PDR + L group confirmed a decrease cell viability (18.75 ± 6.21%), in all probability as a result of the launched DTX had a sure killing impact on most cancers cells. The cell viability of the P-PDR + L group was 30.93 ± 2.11%, which was decrease than that of the M@P-PDR + L group, because the most cancers cell membrane modification may have promoted extra therapeutic brokers to build up within the tumor cells to mediate the therapeutic processes. The cell viabilities of the laser solely group, M@P-PDR solely group and the M@P-DR + L group have been 91.67 ± 6.08%, 91.26 ± 6.70% and 90.43 ± 5.87%, respectively, which weren’t considerably totally different in contrast with that of the management group (95.30 ± 7.30%). Cell injury was additionally analyzed by move cytometry (Fig. 4C), and the outcomes have been according to the CCK-8 outcomes. Moreover, cells after numerous remedies have been additionally stained with CAM/PI to differentiate the dwell (inexperienced fluorescence) and lifeless (purple fluorescence) cells. As proven in Fig. 4B, within the M@P-PDR + L group, nearly all most cancers cells died, exhibiting vibrant purple fluorescence, whereas within the P-PDR + L group, among the most cancers cells appeared inexperienced as a result of lack of environment friendly intracellular uptake. In accordance with the above outcomes, PTT mixed with chemotherapy can inhibit the exercise of tumor cells, and the presence of most cancers cell membranes optimizes the therapeutic impact of tumors.
In vitro therapeutic results of M@P-PDR. A CCK8 outcomes after numerous remedies (n = 3, t-test, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001). B CLSM photos of 4T1 cells co-stained with CAM and PI after numerous remedies to differentiate the dwell (inexperienced fluorescence) and lifeless (purple fluorescence) cells. C Move cytometry outcomes after numerous remedies
Activation of DCs in vitro
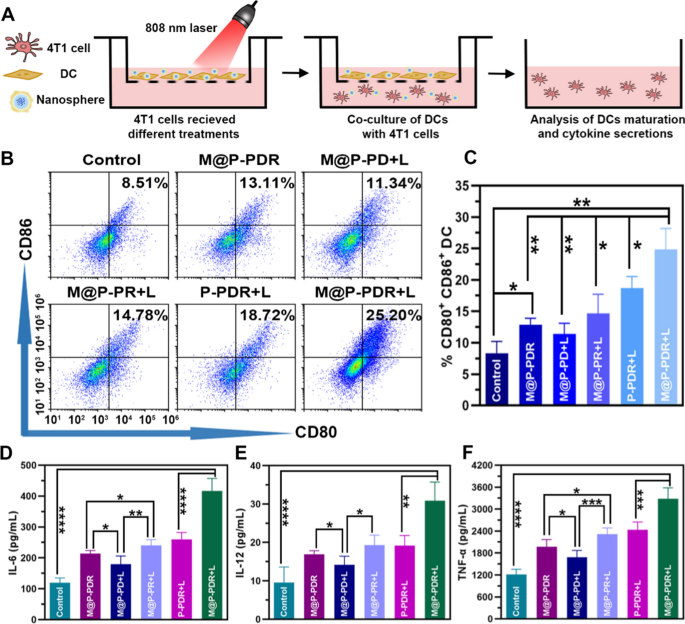
It’s well-known that lifeless or dying cells launch TAAs and damage-associated molecular patterns (DAMPs), corresponding to warmth shock protein 70 (Hsp70) and calreticulin, which might activate antitumor immune responses. These processes are additionally more likely to happen because of the abovementioned therapeutic results. Immune adjuvants corresponding to R837 can additional improve this response. Recognition of R837 by immune cells, corresponding to DCs, that categorical TLR 7 can considerably promote DC maturation and produce a variety of pro-inflammatory cytokines, together with TNF-α (a key marker of mobile immune activation), IL-6 and IL-12 (key markers of innate immunity), thereby stimulating T cell responses [22]. DCs, as probably the most highly effective antigen-presenting cells, play a vital position in activating antitumor immune responses [54]. Basically, DCs seize TAAs launched from lifeless or dying most cancers cells and course of them for presentation on main histocompatibility advanced class I (MHC-I) molecules. Antigen-loaded DCs then migrate from peripheral tissues to the T cell zone of the draining lymph nodes, the place antigen presentation promotes the differentiation of naive T cells into CTLs. Finally, antigen-stimulated T cells go away the lymph nodes and migrate to metastatic tumors, reaching immunotherapy [55, 56]. Notably, solely mature DCs elicit CTLs antitumor responses [57]. The upregulation of typical markers, together with costimulatory molecules (CD11c + , CD80 + , CD86 +), signifies the diploma of DC maturation. Contemplating the sturdy cytotoxic results induced by M@P-PDR, we investigated whether or not M@P-PDR-mediated remedy prompts immune responses in vitro by utilizing a transwell system during which in another way handled 4T1 most cancers cells and untreated bone marrow-derived DCs (naive) have been seeded within the higher and decrease chambers, respectively (Fig. 5A). The maturation efficacy of DCs was measured by move cytometry (Fig. 5B, C). A slight enhance in DC maturation was noticed within the M@P-PDR-treated group, which was in all probability as a result of inevitable launch of a small quantity of R837 from these nanospheres. In comparison with the M@P-PD + L group (with out R837), the extent of DC maturation within the M@P-PDR + L group was drastically elevated, which additional indicated the position of R837 in selling the maturation of DCs. Related cytokines (TNF-α, IL-6 and IL-12) that will be launched by mature DCs have been measured by ELISA. It was discovered that the M@P-PDR group and the M@P-PR + L group confirmed increased secretion ranges than the M@P-PD + L group, which might be attributed to the pivotal position of R837. In comparison with the untargeted P-PDR + L group, DCs within the M@P-PDR + L group secreted rather more cytokines in all probability resulting from homologous focusing on capability mediated by most cancers cell membranes (Fig. 5D–F).
A The design scheme of the transwell system experiment. B–C The expression ranges of CD11c+, CD86+and CD80+on the floor of DCs analyzed by move cytometry after totally different remedies (n = 3). D–F The secretion of IL-6, IL-12 and TNF-α in DC suspensions after totally different remedies (n = 5, t-test, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001)
Biodistribution and in vivo MR/PA bimodal imaging
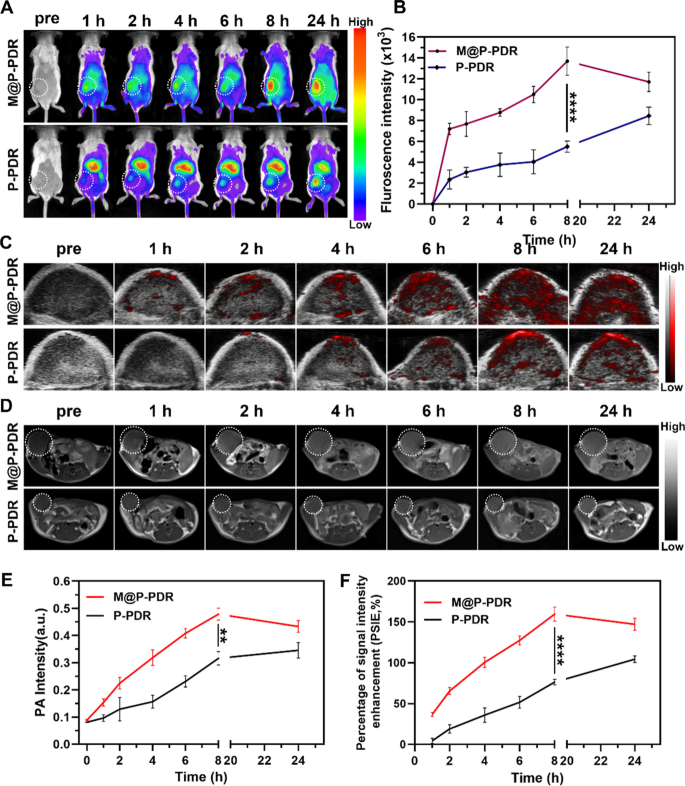
To watch the biodistribution and in vivo focusing on conduct of those M@P-PDR “Nano-targeted cells”, fluorescence imaging of tumor-bearing mice was carried out. DiR-labeled M@P-PDR and P-PDR nanospheres have been intravenously injected, respectively. Within the M@P-PDR-treated group, apparent fluorescence alerts on the tumor websites have been noticed. The alerts elevated with injection time and reached a peak at 8 h (Fig. 6A, B). The imply fluorescence depth of the tumors was 13.70 ± 1.35 × 103, which was 2.49-fold increased than that of the P-PDR-treated group (5.50 ± 0.54 × 103), which may consequence from the homologous focusing on capability of most cancers cell membranes. Extra importantly, important fluorescence alerts have been nonetheless clearly seen at 24 h postinjection, indicating long-term retention of those M@P-PDR “Nano-targeted cells”. Afterward, tumors and main organs (coronary heart, liver, spleen, lung, kidney) have been dissected for ex vivo fluorescence imaging. The fluorescent alerts of tumors within the M@P-PDR group have been evidently stronger than these of the P-PDR group (p < 0.05) (Further file 1: Fig. S10A and B). These outcomes clearly indicated that the most cancers cell membrane-coated nanospheres have been endowed with superior lively focusing on capacity, exhibiting promising prospects for in vivo exact imaging and efficient remedy.
Biodistribution and in vivo MR/PA bimodal imaging of M@P-PDR. A Fluorescence photos of 4T1 tumor-bearing mice at totally different time factors (pre-injection, 1, 2, 4, 6, 8 and 24 h), and B the corresponding fluorescence intensities of tumors (n = 3). C In vivo PA photos of tumors and E the corresponding sign intensities (n = 3). D T1-weighted MR photos and F the corresponding PSIE of tumors (n = 3, t-test, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001)
After intravenous injection, small nanoparticles could also be cleared by the kidneys, such because the graphene nanomaterials (< 50 nm) designed by Omid Akhavan et al. and Zhuang Liu et al. [58, 59], whereas bigger nanoparticles (> 200 nm) might be acknowledged and cleared by the RES of liver and spleen [60, 61]. The nanoparticles we synthesized are bigger in measurement (> 200 nm), so they’re anticipated to be cleared by the RES, which can result in extra aggregation of nanoparticles within the liver than that within the kidney. As well as, after intravenous administration, the exogenous nanoparticles could be nonspecifically intercepted by the liver, which is wealthy in phagocytic cells, leading to substantial aggregation of nanoparticles within the liver. As proven in Further file 1: Fig. S10A and B, the M@P-PDR group had fewer nanoparticles aggregated within the liver, which needs to be ascribed to the decreased clearance of the RES as a result of presence of the antigen retention of the most cancers cell membrane coating.
The aforementioned in vitro experiments confirmed that M@P-PDR nanospheres may act as distinction brokers to reinforce each PA imaging and T1-weighted MR imaging. Due to this fact, bimodal PA and MR imaging efficiency have been additional investigated in vivo. As anticipated, within the M@P-PDR group, the PA alerts inside tumor areas progressively elevated with extended time, and reached a peak at 8 h postinjection (0.479 ± 0.022) as compared with these at preinjection (0.089 ± 0.003) (Fig. 6C, E, Further file 1: S10C). At 24 h postinjection, the PA sign intensities (0.433 ± 0.022) barely decreased as a result of gradual clearance of those nanospheres from tumor tissues. In distinction, within the P-PDR group with out homologous focusing on, the PA alerts have been considerably weaker all through the time course of the remark. The T1-weighted MR imaging outcomes confirmed that the tumors within the M@P-PDR group have been clearly demarcated from the encircling regular tissues with clear anatomical constructions.
As well as, apparent vibrant enhancements have been noticed on the tumor areas over time, reached a peak at 8 h postinjection, and have been sustained for twenty-four h (Fig. 6D, F). PSII was used for quantitative evaluation of T1-weighted MR imaging enhancement. Particularly, the typical T1-weighted sign intensities within the M@P-PDR group elevated by 159.632 ± 8.549% at 8 h postinjection, whereas solely 76.784 ± 3.346% enhancement price was noticed within the P-PDR group. The pseudocolor photos additionally clearly confirmed enhancements (Further file 1: Determine S10D). The pattern of MR imaging is according to that of PA imaging and the enrichment was mirrored by in vivo fluorescence imaging. These outcomes indicated that the floor modification of most cancers cell membranes on P-PDR constructions contributed to the environment friendly accumulation of nanocarriers in tumor websites. Moreover, the wonderful PA/MR bimodal imaging efficiency of those M@P-PDR “Nano-targeted cells” can present a therapeutic time window and information NIR laser irradiation, reaching extra exact remedy supply.
In vivo cocktail remedy analysis
In vivo photothermal efficiency
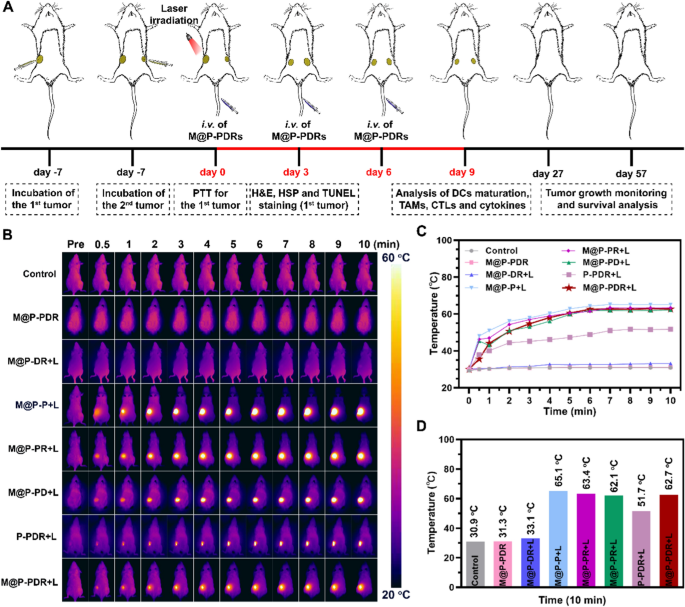
After the enrichment of the PTCAs in tumor areas, the native temperature would rise underneath laser irradiation. The tumors have been uncovered to laser irradiation 8 h after the intravenous injection of nanospheres, and the temperature adjustments have been monitored by an infrared thermal imaging system. As proven in Fig. 7B–D, the temperatures of tumors introduced a slight enhance within the M@P-PDR and M@P-DR + L teams in comparison with the management group within the absence of laser or PB nanoparticles. A major temperature enhance was noticed within the teams with concurrent laser irradiation and PB parts, demonstrating wonderful in vivo photothermal efficiency. The temperature within the M@P-PDR + L group elevated to 62.7 ℃, which was a lot increased than that of the P-PDR + L group (51.7 ℃) with out homologous focusing on capability. Assisted by most cancers cell membrane coating, PTCAs may accumulate in tumors extra effectively to realize extra environment friendly and uniform localized hyperthermia.
In vivo photothermal efficiency of the M@P-PDR “Nano-targeted cells”. A Schematic illustration of the in vivo experimental design. B Infrared thermal photos of 4T1 tumor-bearing Balb/c mice underneath totally different remedy teams. C Photothermal temperature–time curves of the eight teams underneath laser irradiation. and D of the corresponding temperature adjustments at tumor websites throughout irradiation
Immune responses analysis
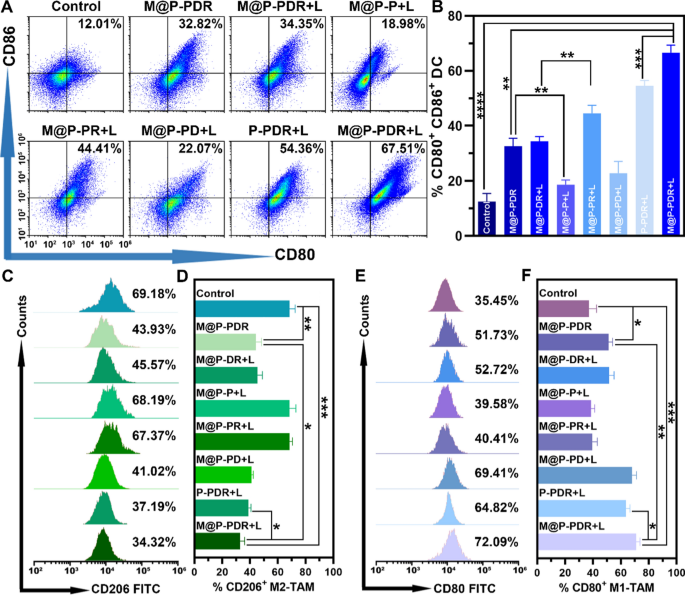
Inspired by the activation of DCs in vitro, the in vivo immune responses have been evaluated subsequent. The experimental design is proven in Fig. 7A. Tumors have been inoculated at each the left and proper mammary fats pads of mice in chronological order, and set as main tumor (1st) and synthetic mimicked metastasis (2nd), respectively. The mice have been randomly divided into eight teams and administered totally different remedies. The day when the remedies designated was set as Day 0. To research the DC maturation degree in vivo, main tumors (1st) (Fig. 8A, B), metastatic tumorS (2nd) (Further file 1: Fig. S11A, B) and lymph node (Further file 1: Fig. S11A, C) have been collected to make single-cell suspensions for move cytometry assay on day 9. Much like the in vitro outcomes, the mixing of the R837 immune adjuvant endowed M@P-PDR “Nano-targeted cells” with a a lot stronger capacity to advertise DC maturation, accompanied by elevated cytokine secretion in vivo. Intimately, the M@P-PDR + L group induced the very best degree of DC maturation (66.56 ± 2.78%), which was considerably increased than the M@P-PD + L group with out R837 (22.81 ± 4.26%), M@P-PDR “Nano-targeted cells” alone (32.65 ± 2.84%), and P-PDR + L group with out homologous focusing on functionality (54.55 ± 1.96%). After PTT mixed with chemotherapy, tumor tissues have been broken, and tumor cell fragments launched TAAs, exhibiting an “autologous most cancers vaccine-like” operate [62]. Particularly within the presence of immune adjuvants, it could promote the maturation of DCs extra effectively [63].
Diploma of in vivo DCs maturation and polarization of TAMs based mostly on M@P-PDR “Nano-targeted cells”. A Move cytometric evaluation of DCs maturation in main tumors (1st) of mice in several remedy teams. and B the corresponding quantification of DCs maturation (n = 3). C Move cytometric evaluation of M2-TAMs (CD206+ F4/80+ CD11b+) in main tumors (1st) and D the corresponding quantification of M2-TAMs (n = 3). E Move cytometric evaluation of M1-TAMs (CD80+ F4/80+ CD11b+) in main tumors (1st) and F the corresponding quantification of M1-TAMs (n = 3, t-test, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001)
Though activation might be established by a number of pathways, the immunosuppressed TME usually ends in a suboptimal immune response. As an essential part of the TME, TAMs play an essential position in tumor immune regulation. As a serious member of the TME, M2-phenotype macrophages promote tumor cell invasion, and metastasis and suppress immune responses by secreting related cytokines (e.g., IL-10), M1-phenotype macrophages counteract tumor progress, and promote inflammatory and immune responses by secreting related cytokines (e.g., IL-6, IL-12 and TNF-α) [64,65,66]. In a pro-immune response pathway, M2-phenotype cancer-promoting TAMs might be repolarized to M1-phenotype cancer-suppressing TAMs underneath sure situations. On this examine, DTX was launched to advertise the polarization of M1-phenotype to M2-phenotype TAMs. To research the polarization of M2-phenotype macrophages, we studied the presence of M1 (F4/80+ CD11b+ CD80+) and M2 (F480+ CD11b+ CD206+) markers on Day 9 after numerous remedies. As proven in Fig. 8C–F and Further file 1: S12A, the expression of F4/80+ CD11b+ CD80+ was considerably upregulated within the teams built-in with DTX, completed by the downregulation of F480+ CD11b+ CD206+ expression, which demonstrated the efficiency of DTX to advertise the polarization of M1-phenotype to M2-phenotype TAMs.
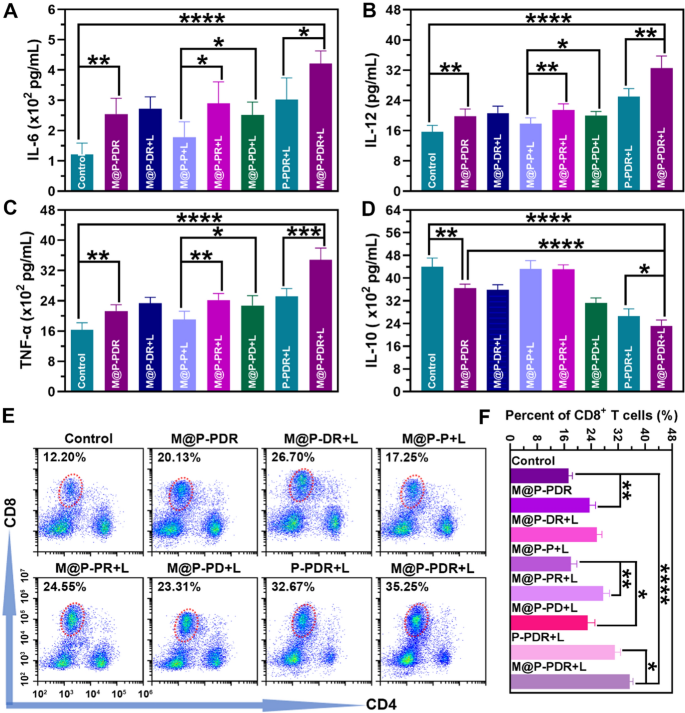
When the host immune standing adjustments, the degrees of cytokines in vivo will change correspondingly. Right here, the degrees of IL-6, IL-12, TNF-α and IL-10 within the eight teams have been investigated by ELISA on Day 9. As proven in Fig. 9A–C, the degrees of those cytokines have been according to the change within the host immune standing (DC maturation and polarization of TAMs) mentioned earlier than. The teams built-in with DTX downregulated the manufacturing of IL-10 (Fig. 9D), additional demonstrating that DTX has a superb capacity to advertise the polarization of M1-phenotype to M2-phenotype TAMs.
In vivo immunostimulatory results based mostly on M@P-PDR “Nano-targeted cells”. A–D The secretion ranges of IL-6, IL-12, TNF-α and IL-10 measured by ELISA assay (n = 5). E Move cytometric evaluation of CD8+ T cell within the spleens of mice in several teams. F Quantification of CD8+ T cells (n = 3, t-test, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001)
CD8+ T cells, particularly CTLs, are important for the anticancer immune response. To judge the T cell response in vivo, the spleens of mice have been collected on Day 9 and T cells within the spleens have been analyzed utilizing move cytometry. The outcomes (Fig. 9E, F) confirmed that the infiltration of CD8+ T cells within the M@P-PDR + L group was 35.50 ± 0.96%, which was considerably increased than that within the management group (17.33 ± 1.13%), the M@P-PDR group (23.54 ± 1.83%), the M@P-DR + L group (25.70 ± 1.57%), the M@P-P + L group (18.01 ± 1.77%), the M@P-PR + L group (27.64 ± 1.86%), the M@P-PD + L group (22.99 ± 2.18%), and the P-PDR + L group (31.09 ± 1.71%), indicating that the processes (PTT, chemotherapy, DC maturation and polarization of TAMs) mediated by M@P-PDR “Nano-targeted cells” triggered wonderful antitumor immune responses. Constantly, immunofluorescence photos of the first and metastatic tumors additionally revealed substantial infiltration of CD8+ T cells (Further file 1: Fig. S12B).
In vivo antitumor remedy for main and metastatic tumors
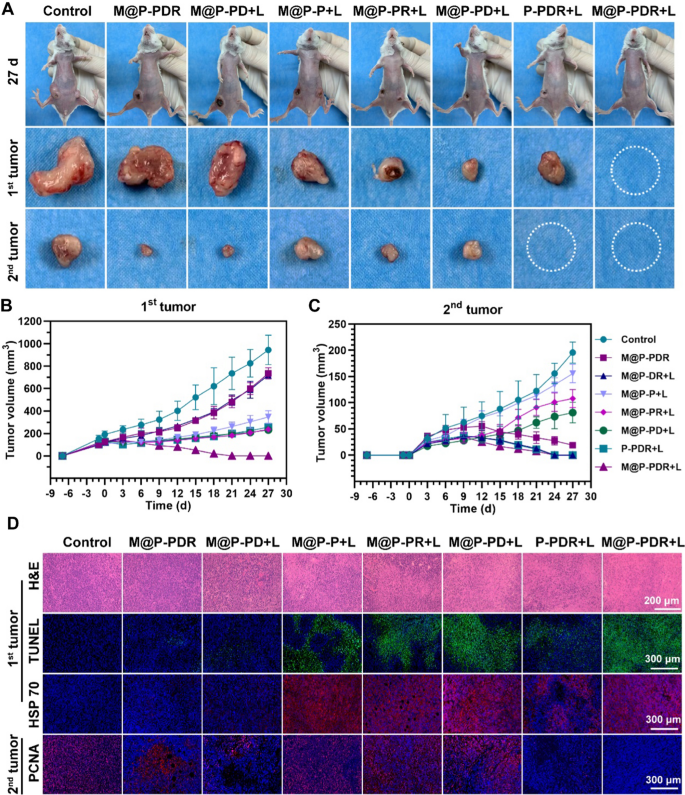
Inspired by the passable immune response, we imagine that the M@P-PDR-based cocktail remedy might be a promising candidate to fight distant tumors. Within the following examine, we investigated whether or not such a powerful immune response initiated by M@P-PDR was accessible for long-term inhibition of metastatic tumors. The therapeutic efficacy was evaluated by monitoring the expansion of main tumors and distant tumors. In contrast with the first tumors (1st) within the management group, all different handled teams exhibited sure inhibitary results on tumor progress (Fig. 10A, B, Further file 1: S13, S14A). Intimately, no important distinction was discovered between the M@P-PDR group and the M@P-DR + L group, whereas restricted tumor progress regression occurred because of the discharge of DTX and R837 in tumor websites. Nonetheless, the photothermal impact of M@P-P drastically inhibited tumor development, with a 3.05-fold enhance compared to the first tumor quantity. Particularly, the first tumors within the M@P-PDR + L group have been remarkably inhibited, suggesting the wonderful antitumor effectivity of such cocktail remedy that concurrently integrates PTT, chemotherapy and immunotherapy. For distant tumor progress in Fig. 10A, B, Further file 1: S13 and S14B, the tumors within the M@P-PDR + L group have been additionally successfully inhibited, which might be ascribed to the sturdy immune response ensuing from R837-induced DC maturation (immune activation) and the DTX-mediated polarization of TAMs (reduction of immunosuppression). To check the therapeutic results extra instantly, the tumors in every group have been collected at 3 d postinjection for TUNEL and HSP70 staining (Fig. 10D). The outcomes confirmed that, within the presence of each PTCAs and laser irradiation, the expression of HSP70 was increased than that in different teams, presenting apparent purple fluorescence. The M@P-PDR + L group confirmed the next expression of HSP70 than the P-PDR + L group, indicating the precise focusing on impact of most cancers cell membranes on the buildup of nanotherapeutic brokers in tumor websites. H&E and TUNEL staining introduced an analogous tendency during which the photothermal results induced large tumor necrosis. The PCNA outcomes of distant tumors revealed that the M@P-PDR + L group exerted in depth antitumor results, with negligible tumor proliferation. Along with the pathological examination, the survival charges of mice in every group have been monitored till Day 57 (Further file 1: Fig. S14C). The mice within the M@P-PDR + L group survived with out apparent tumor recurrence. These outcomes confirmed that the highly effective systemic immune response of M@P-PDR-based cocktail remedy successfully inhibited the expansion of distant tumors, offering a brand new technique for PTT/chemotherapy/immune remedy. H&E staining of main organs (Further file 1: Fig. S15) and the negligible physique weight adjustments (Further file 1: Fig. S16) additional demonstrated the passable biosafety of this synergistic therapeutic modality.
Anti-tumor results of cocktail remedy based mostly on M@P-PDR “Nano-targeted cells”. A Digital pictures of 4T1 tumors on either side in vivo and ex vivo on day 27 after totally different remedies. B Progress curves of the first tumors (1st) and C the distant tumors (2nd) in several teams (n = 5, t-test, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001). D H&E staining, TUNEL staining and HSP70 staining photos of the first tumor (1st) excised at day 3 after totally different remedies, and PCNA staining photos of the distant tumor (2nd) excised at day 9 after totally different remedies
Right here, a comparability with beforehand printed tumor-therapeutic modalities containing these entities summarized right into a desk exhibiting the benefits of this new cocktail therapeutic methods (Further file 1: Desk S1). As one of many conventional most cancers remedy modalities, chemotherapy can inhibit tumor progress to some extent [67, 68]. Nonetheless, most chemotherapeutic medicine have the boundaries of dose-dependent toxicity and unsatisfactory tumor accumulation, which considerably hindered its utility [69]. Some research have demonstrated that DTX can successfully reverse the immunosuppressive TME by polarizing protumoral M2-phenotype TAMs to tumoricidal M1-phenotype TAMs [24]. Immunotherapy has obtained growing consideration resulting from its capacity to activate host defenses to establish, assault, and eradicate most cancers cells [70, 71]. Nonetheless, low immune response price and particular person variations undermine its antitumor efficacy. Furthermore, excessive price and the existence of immune-related opposed reactions (IrAEs) additional restrict its utility [4]. PTT is extensively used for most cancers remedy due to its noninvasiveness, low vitality consumption, and minimal toxicity to regular tissues [72, 73]. Nonetheless, restricted gentle penetration depth could result in incomplete tumor ablation may cause tumor recurrence and even metastasis. Though PTT can activate an immune response that inhibits recurrence and metastasis, the immunosuppressive TME, together with myeloid-derived suppressor cells (MDSCs), prostate most cancers M2-TAMs and regulatory T cells (Tregs), results in ineffective antitumor immune response and immunotherapeutic resistance [4, 74]. On this examine, R837 and DTX was additional built-in into PTT to reinforce the immune response and relieve the immunosuppressive TME. Cocktail remedy not solely successfully ablates the first tumor, but in addition reverses the immunosuppressive TME and enhances the antitumor efficacy.
[ad_2]