[ad_1]
|
Hearken to this text |
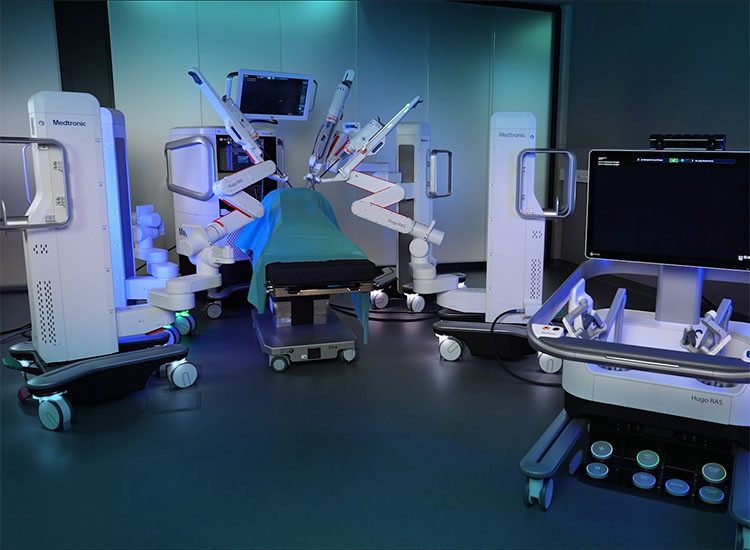
Medtronic has delayed the launch of Hugo, a robot-assisted surgical procedure system, as a result of supply-chain points. | Credit score: Medtronic
Medtronic (NYSE:MDT) Chair & CEO Geoff Martha confirmed that the launch of the Hugo surgical robotic platform is off-schedule.
Hugo acquired CE mark approval final month. The corporate is gearing as much as conduct its FDA investigational system exemption (IDE) scientific trials within the U.S., with extra on its stateside efforts nonetheless to return, Medtronic’s surgical robotics enterprise head Megan Rosengarten advised MDO just lately.
The launch that adopted is at present in what Martha referred to as a “limited-release section” as the corporate focuses on optimizing person expertise because it offers with provide chain and manufacturing points.
“It’s undoubtedly not a requirement concern,” Martha stated on the earnings name. “Demand stays excessive — increased than we will fill at this level.”
The corporate is carrying on with regulatory filings worldwide, working with on U.S. IDE trials; procedures are being carried out with Hugo, Martha stated. One other govt famous on the decision that complexities have come into play, and the corporate ought to have offered extra cushion to expectations for Hugo.
Whereas Medtronic isn’t quantifying projections for subsequent 12 months, the corporate expects a wholesome ramp-up for Hugo, planning for its fiscal 12 months 2023 to “be a powerful 12 months for the robotic.”
“We’re targeted on ensuring the preliminary experiences with surgeons are actually good,” Martha stated. “That’s the rationale for slower income this 12 months, however we anticipate a extremely sturdy ramp subsequent fiscal 12 months.”
Enormous isn’t the one firm off schedule on its surgical robotic system. In October 2021, Johnson & Johnson (J&J) introduced its Ottava surgical robotic system will probably be delayed by two years as a result of technical improvement and supply-chain points.
In November 2020, J&J unveiled the system, claiming that it could provide extra flexibility and management than the rest available on the market. Ottava was initially anticipated to start the verification and validation course of in 2021, and enroll in scientific trials in 2022.
Editor’s Observe: This text first appeared on our sister web site Medical Design and Outsourcing.
[ad_2]

