[ad_1]
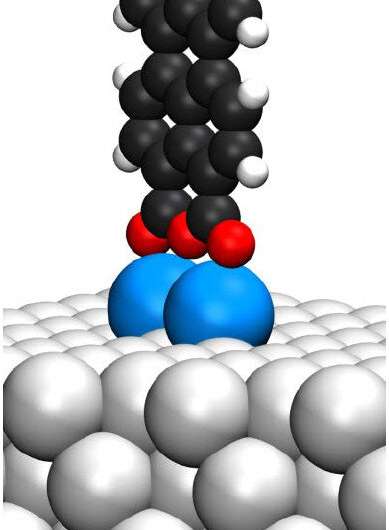
Nanoscale equipment has many makes use of, together with drug supply, single-atom transistor know-how, or reminiscence storage. Nevertheless, the equipment have to be assembled on the nanoscale, which is a substantial problem for researchers.
For nanotechnology engineers the last word objective is to have the ability to assemble purposeful equipment part-by-part on the nanoscale. Within the macroscopic world, we will merely seize objects to assemble them. It’s not not possible to “seize” single molecules anymore, however their quantum nature makes their response to manipulation unpredictable, limiting the power to assemble molecules one after the other. This prospect is now a step nearer to actuality, because of a world effort led by the Analysis Centre Jülich of the Helmholtz society in Germany, together with researchers from the Division of Chemistry on the College of Warwick.
Within the paper, “The stabilization potential of a standing molecule,” revealed immediately, 10 November 2021 within the journal Science Advances, a world group of researchers has been capable of reveal the generic stabilization mechanism of a single standing molecule, which can be utilized within the rational design and development of three-dimensional molecular units at surfaces.
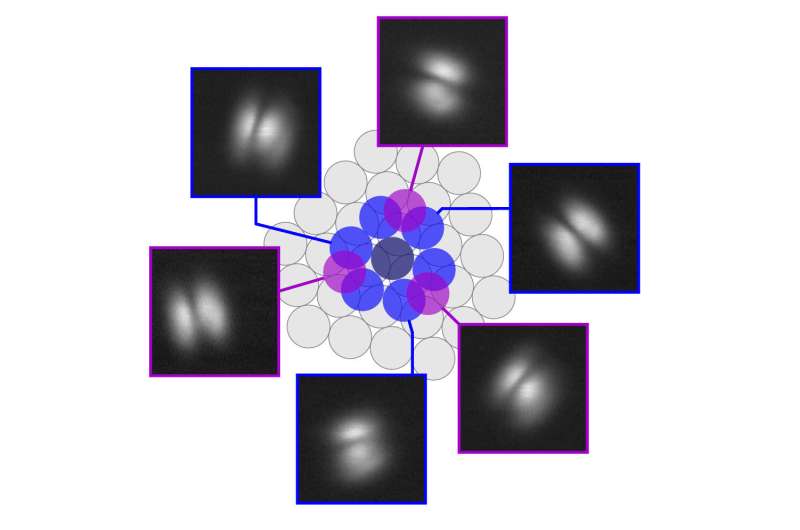
The scanning probe microscope (SPM) has introduced the imaginative and prescient of molecular-scale fabrication nearer to actuality, as a result of it gives the potential to rearrange atoms and molecules on surfaces, thereby permitting the creation of metastable buildings that don’t type spontaneously. Utilizing SPM, Dr. Christian Wagner and his group had been capable of work together with a single standing molecule, perylene-tetracarboxylic dianhydride (PTCDA) on a floor to check the thermal stability and temperature at which the molecule would stop to be steady and would drop again into its pure state the place it adsorbs flat on the floor. This temperature stands at -259.15 Celsius, solely 14 levels above absolutely the zero-temperature level.
Quantum chemical calculations carried out in collaboration with Dr. Reinhard Maurer from the Division of Chemistry on the College of Warwick had been capable of reveal that the refined stability of the molecule stems from the competitors of two sturdy counteracting quantum forces, specifically the long-range attraction from the floor and the short-range restoring power arising from the anchor level between molecule and the floor.
Dr. Reinhard Maurer from the Division of Chemistry on the College of Warwick feedback, “The stability of interactions that retains the molecule from falling over could be very refined and a real problem for our quantum chemical simulation strategies. Along with instructing us in regards to the elementary mechanisms that stabilize such uncommon nanostructures, the challenge additionally helped us to evaluate and enhance the capabilities of our strategies.”
Dr. Christian Wagner from the Peter Grünberg Institute for Quantum Nanoscience (PGI-3) at Analysis Centre Jülich feedback, “To make technological use of the fascinating quantum properties of particular person molecules, we have to discover the fitting stability: They have to be immobilized on a floor, however with out fixing them too strongly, in any other case they’d lose these properties. Standing molecules are best in that respect. To measure how steady they really are, we needed to stand them up again and again with a pointy steel needle and time how lengthy they survived at totally different temperatures.”
Now that the interactions that give rise to a steady standing molecule are recognized, future analysis can work in direction of designing higher molecules and molecule-floor hyperlinks to tune these quantum interactions. This will help to extend stability and the temperature at which molecules will be switched into standing arrays in direction of workable situations. This raises the prospect of nanofabrication of equipment on the nanoscale.
Marvin Knol et al, The stabilization potential of a standing molecule, Science Advances (2021). DOI: 10.1126/sciadv.abj9751
Quotation:
Competing quantum interactions allow single molecules to face up (2021, November 10)
retrieved 10 November 2021
from https://phys.org/information/2021-11-quantum-interactions-enable-molecules.html
This doc is topic to copyright. Aside from any honest dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for data functions solely.
[ad_2]

