[ad_1]
The whole lot of the identified universe is teeming with an infinite variety of molecules. However what fraction of those molecules have potential drug-like traits that can be utilized to develop life-saving drug remedies? Thousands and thousands? Billions? Trillions? The reply: novemdecillion, or 1060. This gargantuan quantity prolongs the drug growth course of for fast-spreading ailments like Covid-19 as a result of it’s far past what present drug design fashions can compute. To place it into perspective, the Milky Approach has about 100 thousand million, or 108, stars.
In a paper that might be offered on the Worldwide Convention on Machine Studying (ICML), MIT researchers developed a geometrical deep-learning mannequin known as EquiBind that’s 1,200 instances sooner than one of many quickest present computational molecular docking fashions, QuickVina2-W, in efficiently binding drug-like molecules to proteins. EquiBind is predicated on its predecessor, EquiDock, which makes a speciality of binding two proteins utilizing a method developed by the late Octavian-Eugen Ganea, a latest MIT Laptop Science and Synthetic Intelligence Laboratory and Abdul Latif Jameel Clinic for Machine Studying in Well being (Jameel Clinic) postdoc, who additionally co-authored the EquiBind paper.
Earlier than drug growth may even happen, drug researchers should discover promising drug-like molecules that may bind or “dock” correctly onto sure protein targets in a course of generally known as drug discovery. After efficiently docking to the protein, the binding drug, also referred to as the ligand, can cease a protein from functioning. If this occurs to an important protein of a bacterium, it could possibly kill the bacterium, conferring safety to the human physique.
Nevertheless, the method of drug discovery may be pricey each financially and computationally, with billions of {dollars} poured into the method and over a decade of growth and testing earlier than last approval from the Meals and Drug Administration. What’s extra, 90 % of all medicine fail as soon as they’re examined in people resulting from having no results or too many unintended effects. One of many methods drug corporations recoup the prices of those failures is by elevating the costs of the medicine which might be profitable.
The present computational course of for locating promising drug candidate molecules goes like this: most state-of-the-art computational fashions rely on heavy candidate sampling coupled with strategies like scoring, rating, and fine-tuning to get one of the best “match” between the ligand and the protein.
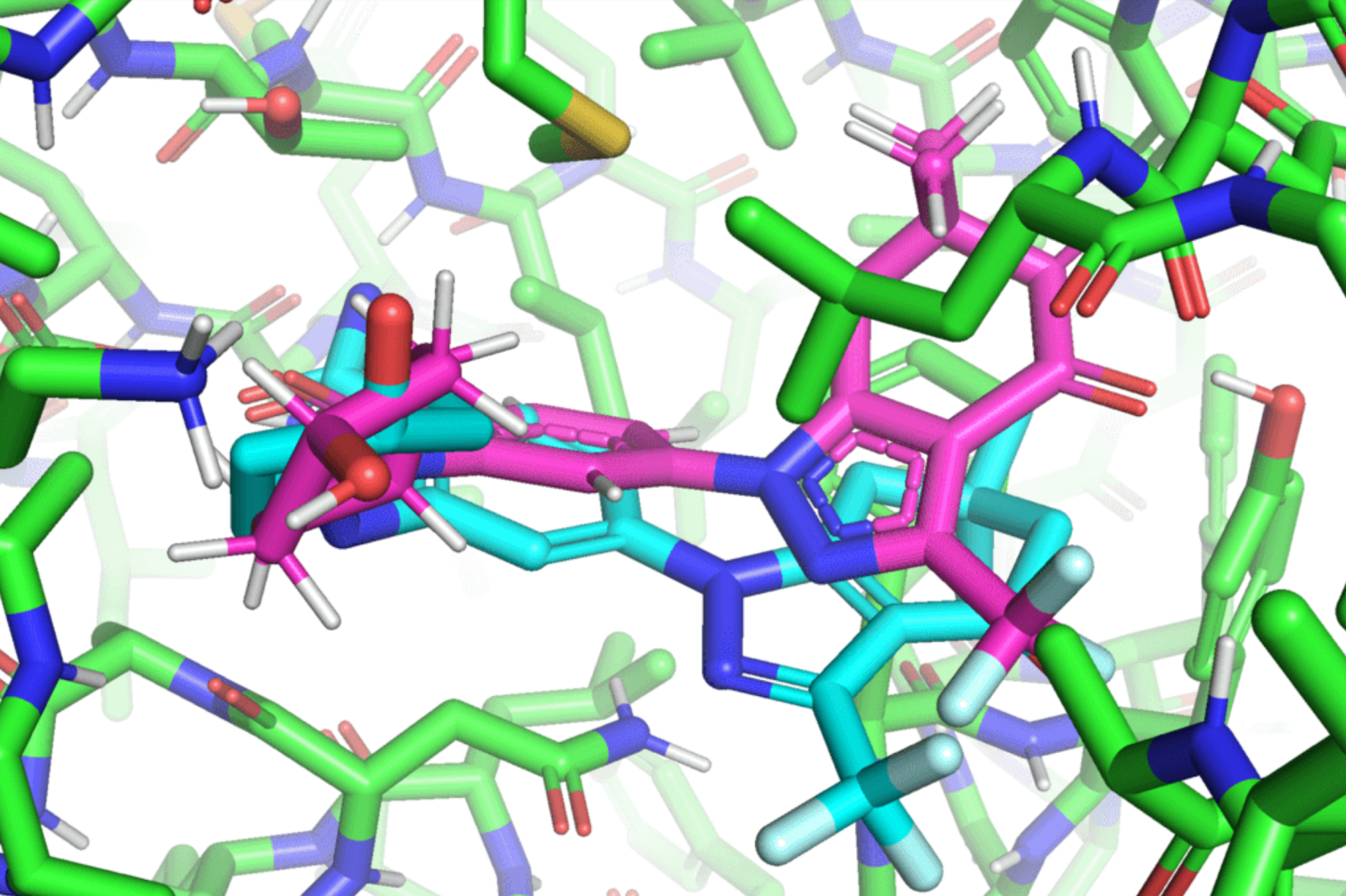
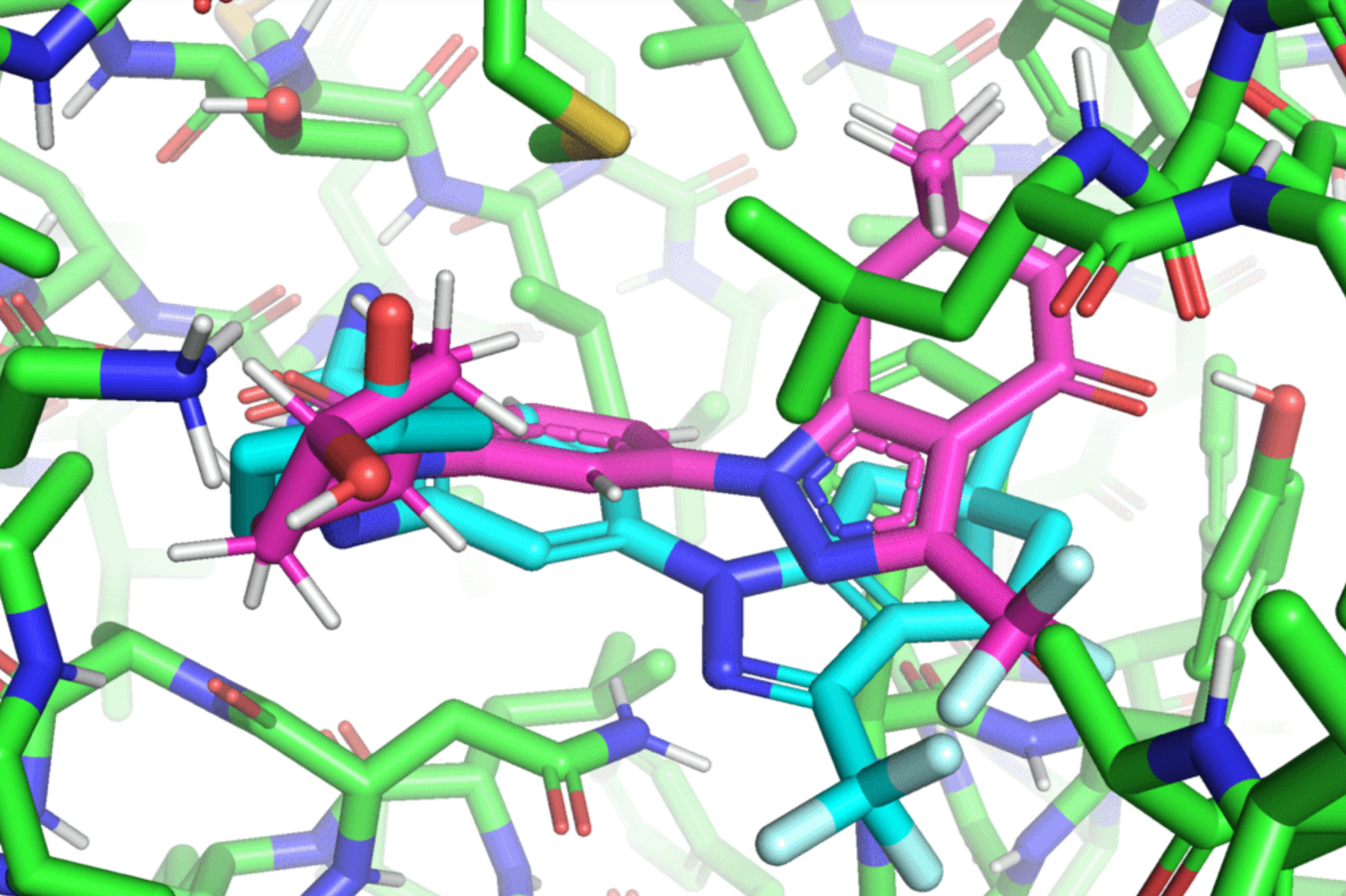
Hannes Stärk, a first-year graduate scholar on the MIT Division of Electrical Engineering and Laptop Science and lead creator of the paper, likens typical ligand-to-protein binding methodologies to “making an attempt to suit a key right into a lock with a number of keyholes.” Typical fashions time-consumingly rating every “match” earlier than selecting one of the best one. In distinction, EquiBind straight predicts the exact key location in a single step with out prior information of the protein’s goal pocket, which is called “blind docking.”
In contrast to most fashions that require a number of makes an attempt to discover a favorable place for the ligand within the protein, EquiBind already has built-in geometric reasoning that helps the mannequin study the underlying physics of molecules and efficiently generalize to make higher predictions when encountering new, unseen information.
The discharge of those findings shortly attracted the eye of trade professionals, together with Pat Walters, the chief information officer for Relay Therapeutics. Walters steered that the staff strive their mannequin on an already present drug and protein used for lung most cancers, leukemia, and gastrointestinal tumors. Whereas many of the conventional docking strategies didn’t efficiently bind the ligands that labored on these proteins, EquiBind succeeded.
“EquiBind gives a singular answer to the docking drawback that comes with each pose prediction and binding website identification,” Walters says. “This strategy, which leverages info from hundreds of publicly accessible crystal constructions, has the potential to affect the sector in new methods.”
“We had been amazed that whereas all different strategies obtained it utterly fallacious or solely obtained one right, EquiBind was capable of put it into the proper pocket, so we had been very completely satisfied to see the outcomes for this,” Stärk says.
Whereas EquiBind has acquired a substantial amount of suggestions from trade professionals that has helped the staff think about sensible makes use of for the computational mannequin, Stärk hopes to seek out totally different views on the upcoming ICML in July.
“The suggestions I’m most wanting ahead to is strategies on how one can additional enhance the mannequin,” he says. “I wish to focus on with these researchers … to inform them what I believe may be the subsequent steps and encourage them to go forward and use the mannequin for their very own papers and for their very own strategies … we’ve had many researchers already reaching out and asking if we predict the mannequin may very well be helpful for his or her drawback.”
This work was funded, partially, by the Pharmaceutical Discovery and Synthesis consortium; the Jameel Clinic; the DTRA Discovery of Medical Countermeasures In opposition to New and Rising threats program; the DARPA Accelerated Molecular Discovery program; the MIT-Takeda Fellowship; and the NSF Expeditions grant Collaborative Analysis: Understanding the World By means of Code.
This work is devoted to the reminiscence of Octavian-Eugen Ganea, who made essential contributions to geometric machine studying analysis and generously mentored many college students — a superb scholar with a humble soul.
[ad_2]