| Feb 14, 2022 |
|
|
|
(Nanowerk Information) A brand new sustainable and sensible technique for producing hydrogen from water has been found by a workforce of researchers on the RIKEN Middle for Sustainable Useful resource Science (CSRS) in Japan led by Ryuhei Nakamura.
|
|
Not like present strategies, the brand new technique doesn’t require uncommon metals which can be costly or in brief provide. As an alternative, hydrogen for gas cells and agricultural fertilizers can now be produced utilizing cobalt and manganese, two pretty widespread metals.
|
|
The research was printed in Nature Catalysis (“Enhancing the soundness of cobalt spinel oxide in direction of sustainable oxygen evolution in acid”).
|
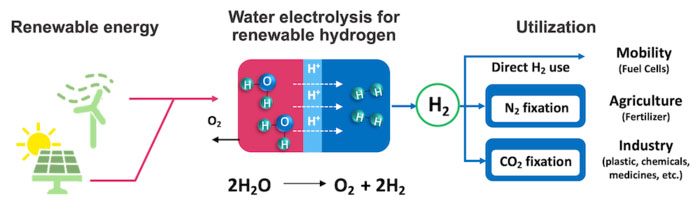 |
| This schematic exhibits the idea for sustainable hydrogen manufacturing. Electrical energy from renewable sources (photo voltaic, wind) is used to separate water into oxygen and hydrogen (electrolysis). The hydrogen can then be used for as gas, to assist make fertilizer from ammonia, and in different industries. (Picture: RIKEN)
|
|
Not like standard fossil fuels that generate carbon dioxide upon combustion, hydrogen is a clear gas that solely produces water as a byproduct. If hydrogen will be extracted from water utilizing renewable electrical energy, the power grid will be made clear, renewable, and sustainable. Moreover, hydrogen is the important thing ingredient wanted to provide ammonia, which is utilized in nearly all artificial fertilizers. However as a substitute of cleanly extracting hydrogen from water, presently, ammonia crops use fossil fuels to provide the hydrogen they want.
|
|
So why are we nonetheless utilizing fossil fuels? One motive is that the hydrogen extraction course of itself—electrolysis—is pricey and never but sustainable.
|
|
“That is primarily because of a scarcity of excellent catalysts,” says Nakamura. “Along with with the ability to face up to the cruel acidic setting, the catalyst have to be very energetic. If not, the quantity of electrical energy wanted for the response to provide a given quantity of hydrogen soars, and with it, so does the associated fee.”
|
|
At present, essentially the most energetic catalysts for water electrolysis are uncommon metals like platinum and iridium, which creates a dilemma as a result of they’re costly and thought of “endangered species” amongst metals. Switching the entire planet to hydrogen gas proper now would require about 800 years’ price of iridium manufacturing, an quantity which could not even exist. Alternatively, plentiful metals akin to iron and nickel should not energetic sufficient and have a tendency to dissolve instantly within the harsh acidic electrolysis setting.
|
|
Of their seek for a greater catalyst, the researchers checked out combined cobalt and manganese oxides. Cobalt oxides will be energetic for the required response, however corrode in a short time within the acidic setting. Manganese oxides are extra steady, however should not practically energetic sufficient. By combining them, the researchers hoped to benefit from their complimentary properties. Additionally they needed to think about the excessive present density wanted for sensible software exterior the laboratory. “For industrial scale hydrogen manufacturing, we would have liked to set our research’s goal present density to about 10 to 100 occasions increased than what has been utilized in previous experiments,” says co-first writer Shuang Kong. “The excessive currents led to quite a few issues akin to bodily decomposition of the catalyst.”
|
|
Finally, the workforce overcame these points by trial and error, and found an energetic and steady catalyst by inserting manganese into the spinel lattice of Co3O4, producing the combined cobalt manganese oxide Co2MnO4.
|
|
Testing confirmed that Co2MnO4 carried out very effectively. Activation ranges have been near these for state-of-the artwork iridium oxides. Moreover, the brand new catalyst lasted over two months at a present density of 200 milliamperes per sq. centimeter, which might make it efficient for sensible use. In contrast with different non-rare metallic catalysts, which generally final solely days or even weeks at a lot decrease present densities, the brand new electrocatalyst could possibly be a recreation changer.
|
|
“We’ve achieved what has eluded scientists for many years,” says co-first writer Ailong Li. “Hydrogen manufacturing utilizing a extremely energetic and steady catalyst produced from plentiful metals. In the long term, we imagine that it is a enormous step in direction of making a sustainable hydrogen economic system. Like different renewable applied sciences akin to photo voltaic cells and wind energy, we count on the price of inexperienced hydrogen expertise to plummet within the close to future as extra advances are made.”
|
|
The subsequent step in lab will likely be to seek out methods to increase the lifetime of the brand new catalyst and improve its exercise ranges much more. “There may be all the time room for enchancment,” says Nakamura, “and we proceed to attempt for a non-rare metallic catalyst that matches the efficiency of present iridium and platinum catalysts.”
|

